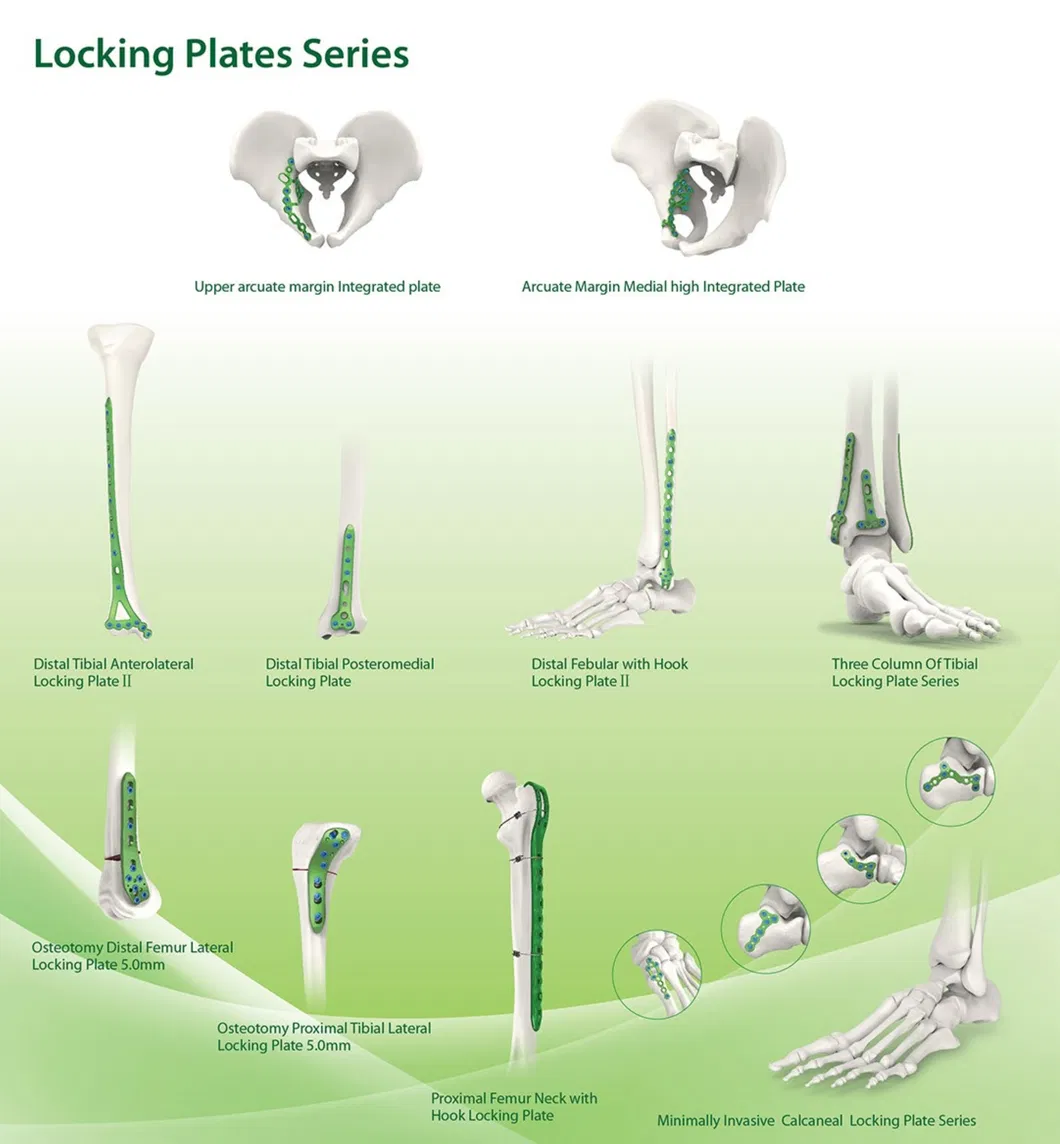
📋 Basic Information
Certification
CE, ISO13485
🔍 Product Description
| Products Name |
NO. |
Specification |
| Straight Maxillofacial Plate, Type-IV |
1500-0115 |
6 holes (Interspace:6.0mm) |
| 1500-0116 |
6 holes (Interspace:7.5mm) |
| 1500-0117 |
6 holes (Interspace:9.0mm) |
| 1500-0118 |
6 holes (Interspace:12mm) |
| Compatibility: Use with 1.5mm Self-tapping Screw or 1.5mm Self-drilling Screw |
🏢 Profile
Established in 1958, we are engaged in the development, manufacture and sales of orthopedic implants and instruments. As a director company of the Medical Apparatus Industry Association, our product line covers Spine pedical screws, Interlocking nails, Locking plates/screws, trauma plates/screws, cannulated screws, external fixators, hip prosthesis and surgical instruments.
Our products are characterized with multi-varieties and complete specifications. All medical apparatus have passed the approval and authentication of national and provincial superintendent offices. High-quality production is supported by advanced equipment, including imported CNC, high precision digital control machines, and a 100,000 Grade axenic purification chamber.
We adhere to the development tenet of "create high quality with science and technology, serve the society with love". We pursue perfect design, innovative technology, and top-ranking products combined with complete after-sales service.
❓ FAQ
What materials are used for these maxillofacial plates?
The plates are manufactured using high-grade Titanium, ensuring biocompatibility and strength for maxillofacial procedures.
Which quality certifications do your products carry?
All products are produced according to international standards and hold CE, ISO 9001, and ISO 13485 certifications.
How do you ensure the quality of the implants before shipping?
We implement a strict quality control system, including pre-production samples and a final 100% inspection before shipment.
What is your monthly production capacity for orthopedic implants?
Our current production capacity is approximately 10,000 pieces per month, supported by high-precision CNC equipment.
What types of shipping and payment terms are accepted?
We accept delivery via DHL, TNT, EMS, UPS, FedEx, and Air. Payment terms include T/T, L/C, Western Union, and Escrow.
Can you provide OEM or ODM services for specialized requirements?
Yes, we provide OEM and ODM services, allowing for customized branding and specifications based on clinical needs.