Basic Specifications
Certification
CE, ISO13485
Material
Peek, Titanium Screw
Function
Connect Soft Tissue and Bone
Product Description
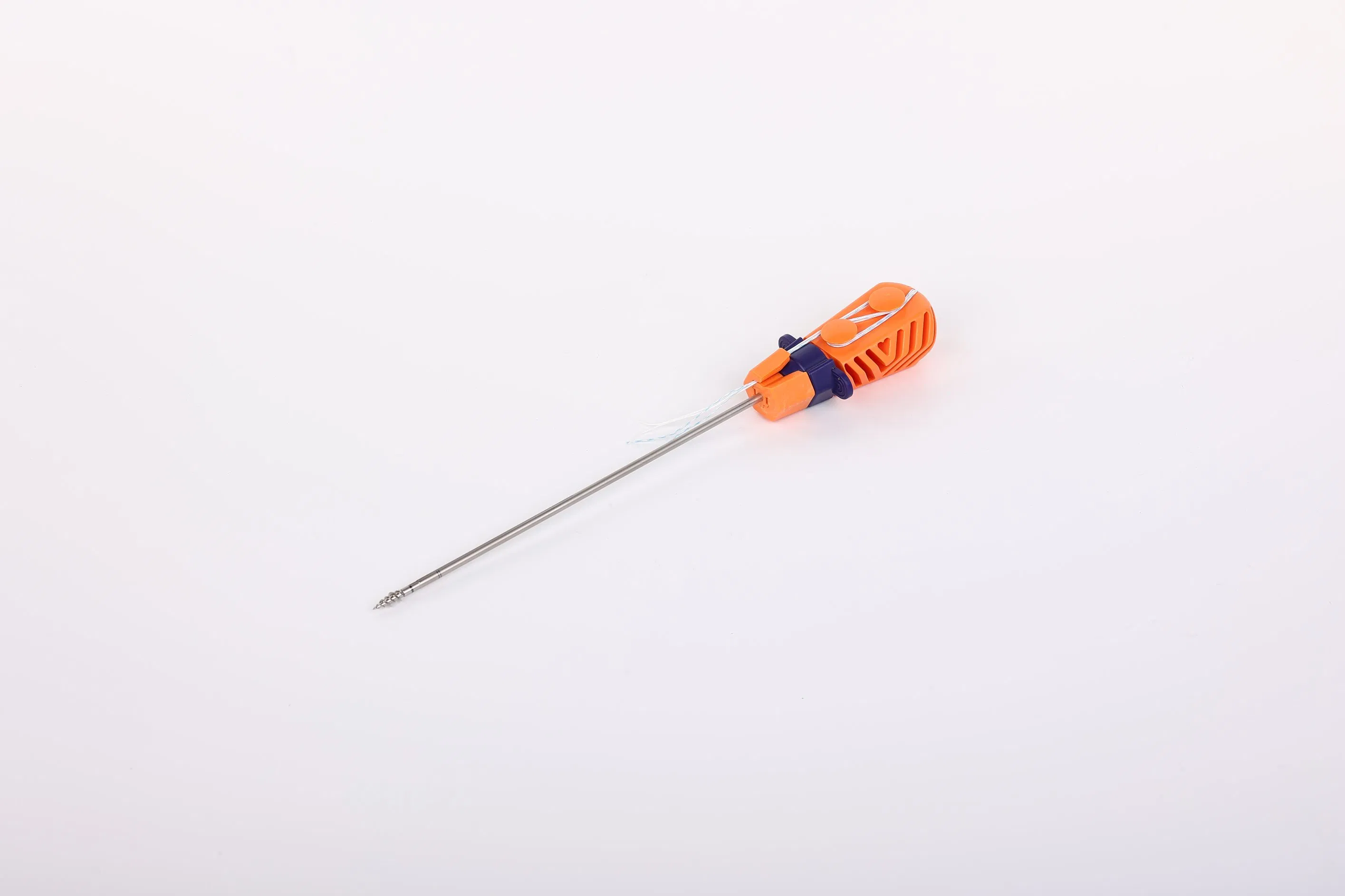
System Composition: The Suture Anchor System consists of an anchor, nonabsorbable suture, and an inserter.
Premium Materials: The anchor is constructed from Ti6Al4V alloy, strictly meeting ISO 5832-3:2016 requirements.
High-Performance Suture: Made of ultra-high molecular weight polyethylene (UHMWPE) without coating, woven for maximum strength (compliant with ASTM F2848-17).
Ergonomic Inserter: The contact part is stainless steel (ASTM F899-12b), featuring a polycarbonate and ABS handle for precise control.
Professional Capabilities
Comprehensive orthopedic solutions including Suture Anchor Systems, Meniscal Repair Systems, Shaver Blades, Bone Cement Mixers, Rib Splints, and Finger Splints. All production is conducted in Class 100,000 clean workshops with Class 10,000 purification laboratories to ensure the highest quality and safety standards.
Frequently Asked Questions
What materials are used for the anchors?
The anchors are manufactured from Ti6Al4V Titanium alloy and are also available in Peek materials to ensure biocompatibility and strength.
What is the shelf life and sterilization method?
The product is EO (Ethylene Oxide) sterilized and comes with a 3-year warranty/shelf life under proper storage conditions.
Are there different sizes available for various surgeries?
Yes, we provide multiple sizes including 1.8mm, 2.8mm, 3.5mm, 4.0mm, 4.5mm, 5.0mm, 5.5mm, and 6.5mm to accommodate different orthopedic requirements.
Does the product meet international medical standards?
Absolutely. All products are fully compliant with CE certification and ISO 13485 standards for global medical use.
Do you support custom OEM requirements?
Yes, we offer professional OEM services, including customized designs and packaging solutions to meet specific brand needs.
Is the suture material safe for human implantation?
Yes, the suture is made of UHMWPE material. The blue dye used is FDA-listed and approved for medical implantation.