


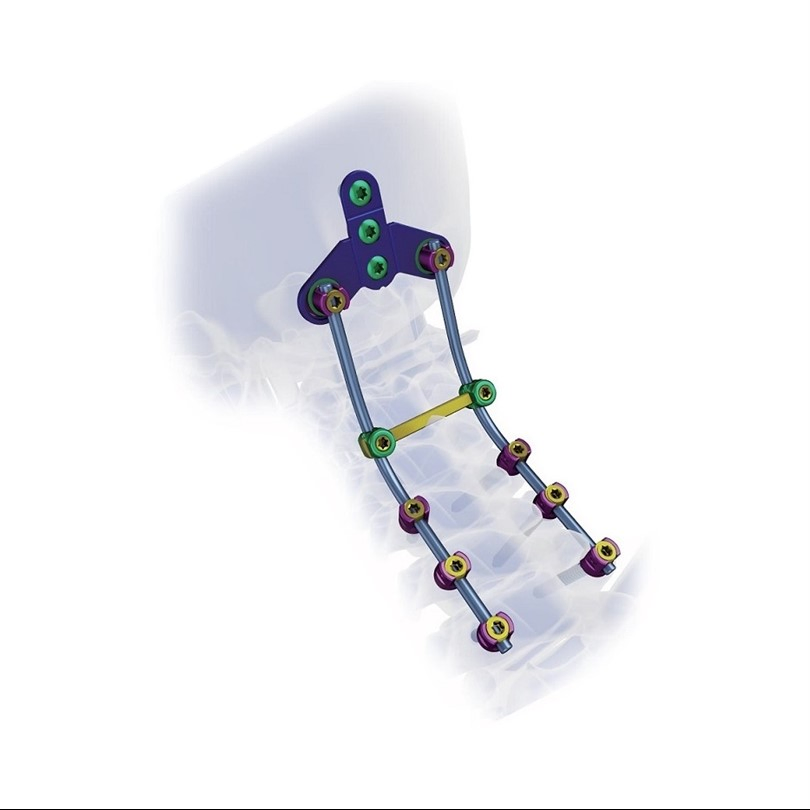
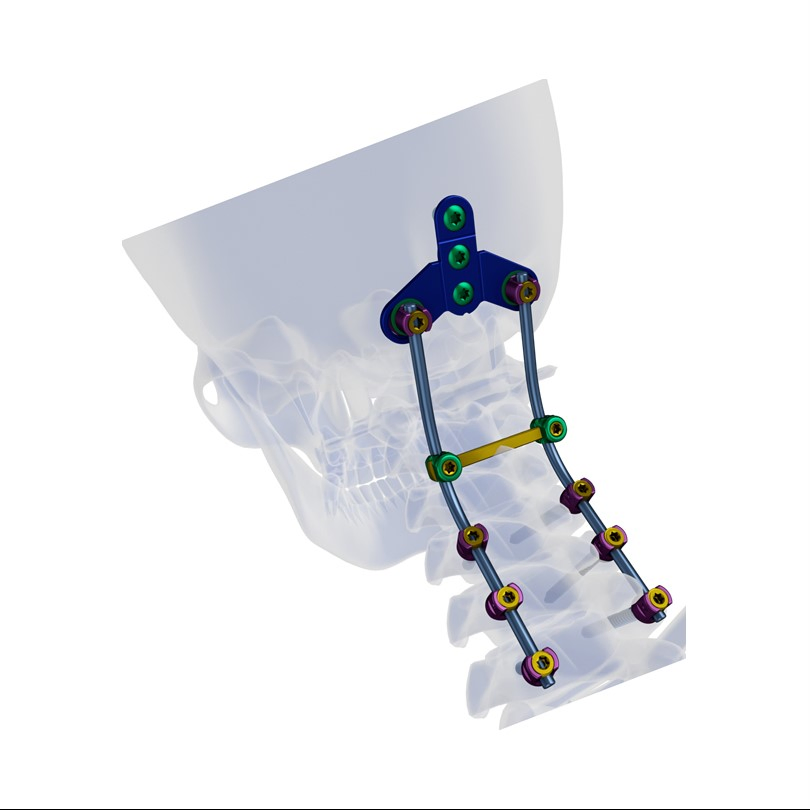
1 / 5










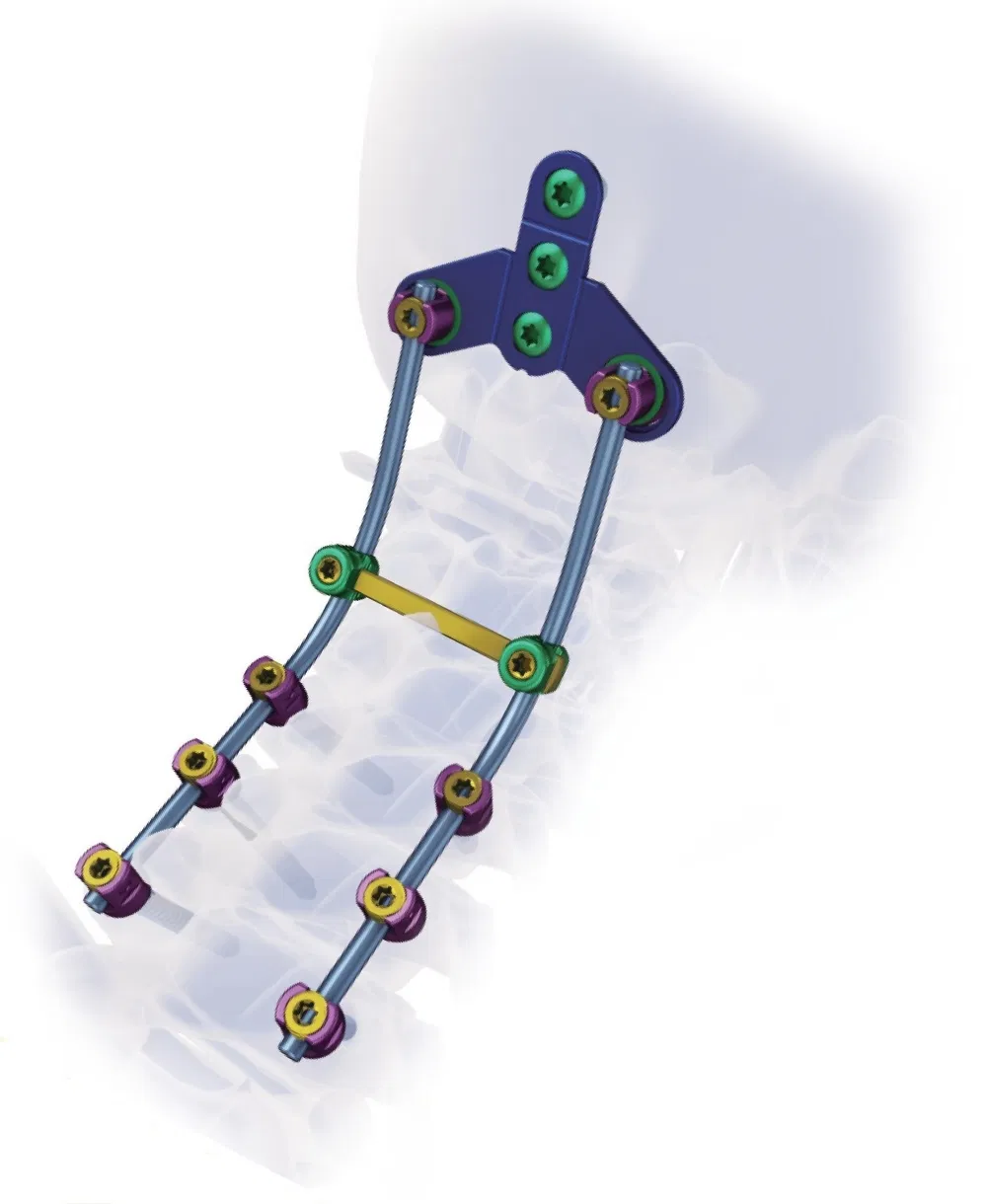
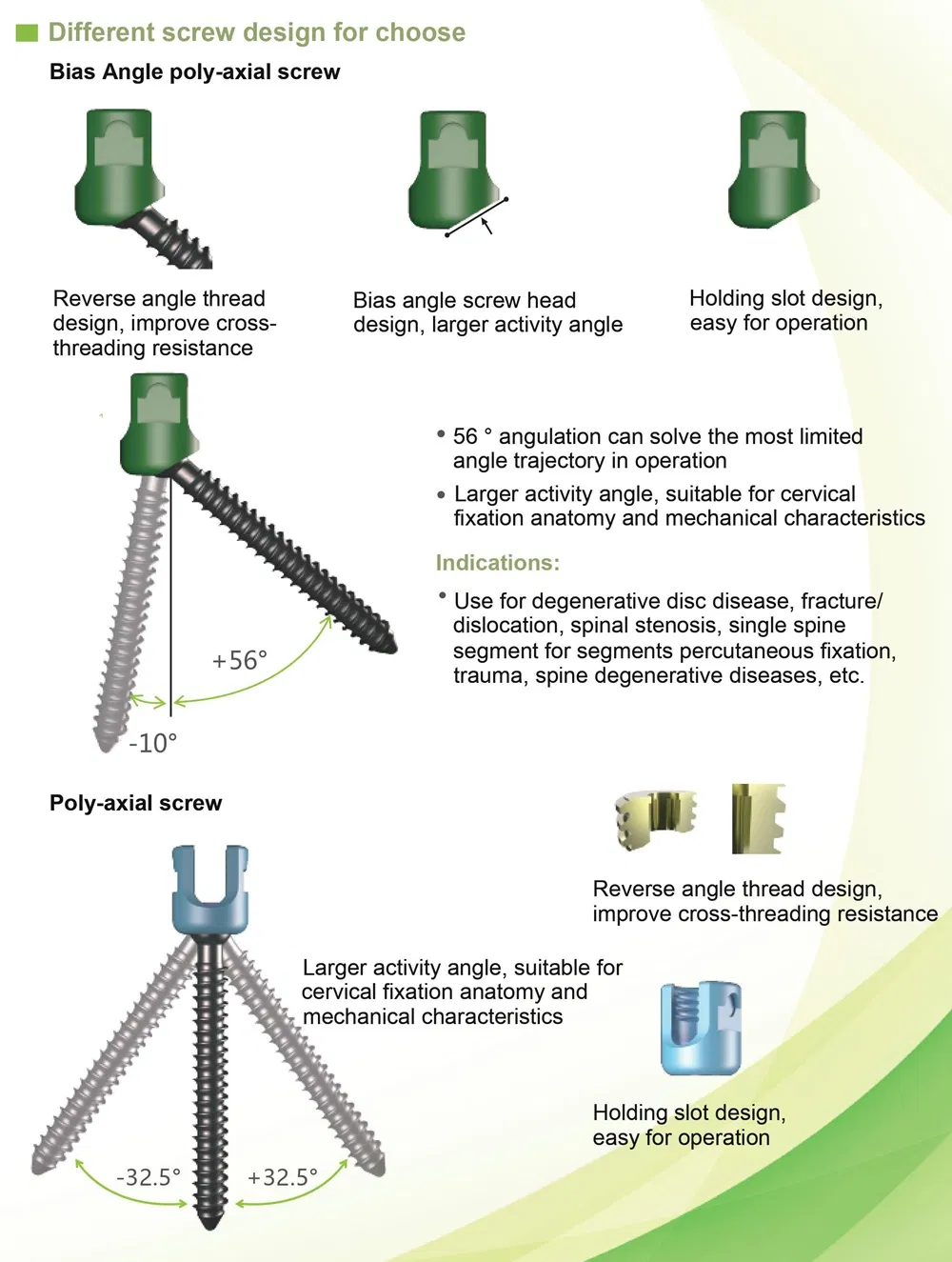


- Implants: Inner plastic bag + outer carton box. Screws packed in sets of 5; plates in individual waterproof bags.
- Instruments Set: Inner instruments box + outer carton box.
- Customized packing design service available.
- International Express: DHL, UPS, FedEx, etc.
- Air transportation for rapid supply.



