M
Manufacturing Excellence

All products are certified by CE & ISO13485. The system combines convenience and power for the removal of contaminated water, necrotic tissue, bone debris, wound pollutants, and foreign material whilst providing controlled pressure for efficient cleansing and irrigation on the site of flat wound surfaces, osteotomy surfaces, or the intra-medullary.
Products are continuously improved via clinical research and feedback from surgeons. Noise and weight (currently only 772g) are minimized to ensure favorable handling during procedures. It is an essential medical instrument in modern orthopedic surgeries.
?
Frequently Asked Questions
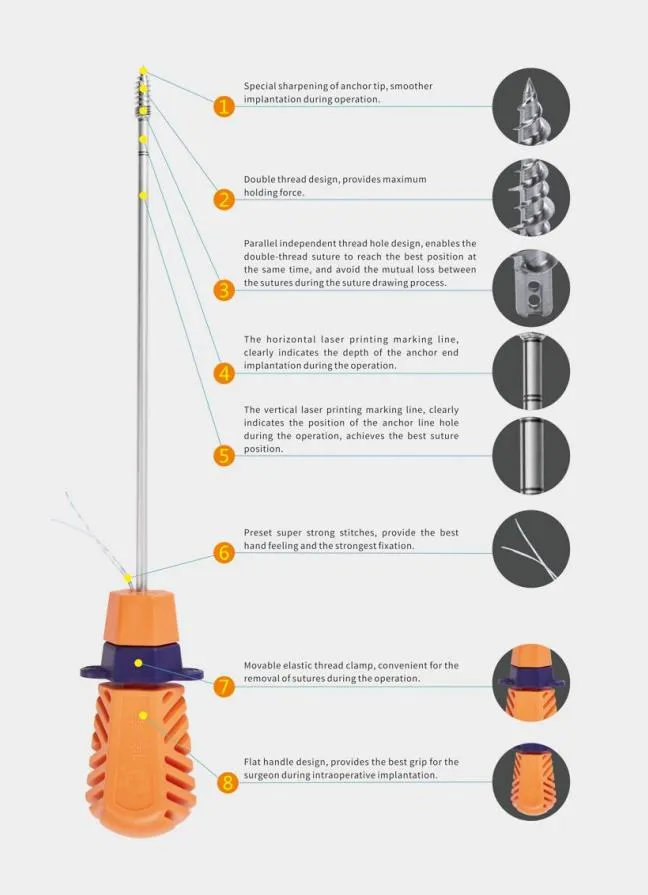
What is the primary material used for the suture anchors?
The suture anchors are made of high-quality Titanium, which is non-bioabsorbable and ideal for orthopedic surgical procedures.
Is the suture anchor reusable?
No, this product is designed as a disposable medical device for single-use to ensure safety and hygiene.
What certifications does this product hold?
The product is fully certified with CE and ISO13485 standards, meeting international medical quality requirements.
What is the shelf life and sterilization method?
The product has a validity period of 3 years and is sterilized using the Eo (Ethylene Oxide) sterilization method.
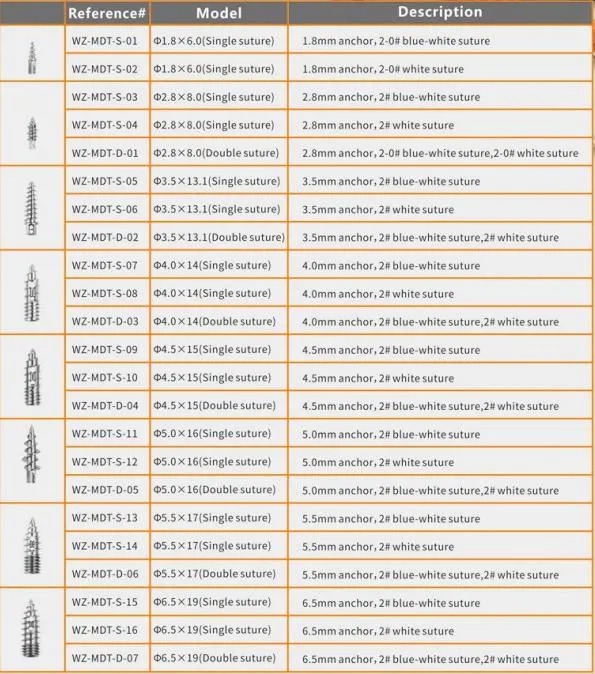
What sizes are available for the suture anchors?
We offer a wide range of specifications including 1.8mm, 2.8mm, 3.5mm, 4.0mm, 4.5mm, 5.0mm, 5.5mm, and 6.5mm.
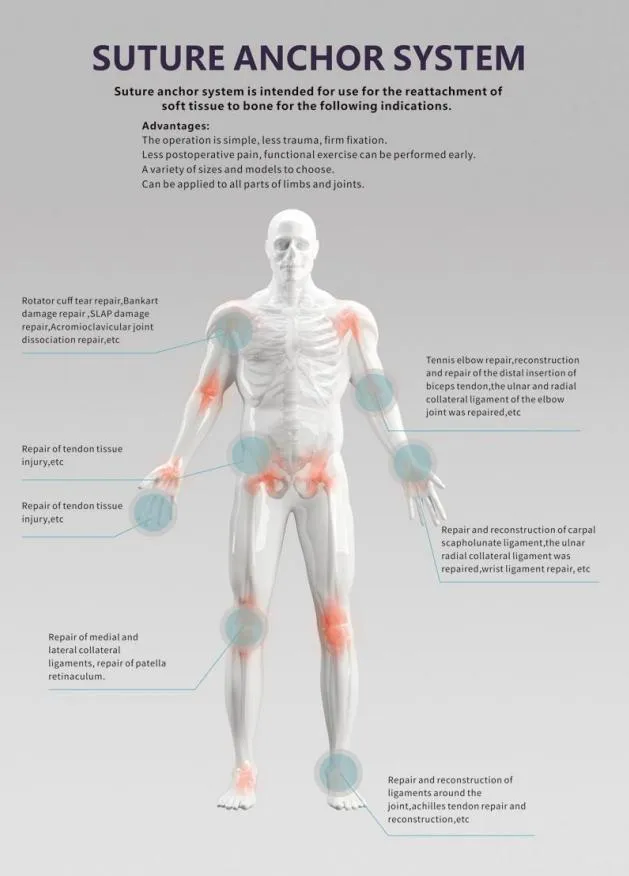
What is the intended use of this medical device?
It is primarily used in orthopedic surgery to connect soft tissue and bone, providing a secure and stable fixation.