










| Model NO. | W03 |
|---|---|
| Material | Aluminum/Ss |
| Feature | Disposable |
| Certification | CE, ISO13485 |
| Group | Adult |
| Usage | Bone Fracture Fixation |
| MOQ | 1 PC |
| Color | Black |
| Shipping | DHL\UPS\FedEx\EMS\TNT Air Cargo |
| OEM/ODM | Accept |
| Transport Package | PE Bags, One Inner Carton and One Outer Carton. |
| HS Code | 9021100000 |
| Production Capacity | 1000PCS/Month |
| Number | W03 |
|---|---|
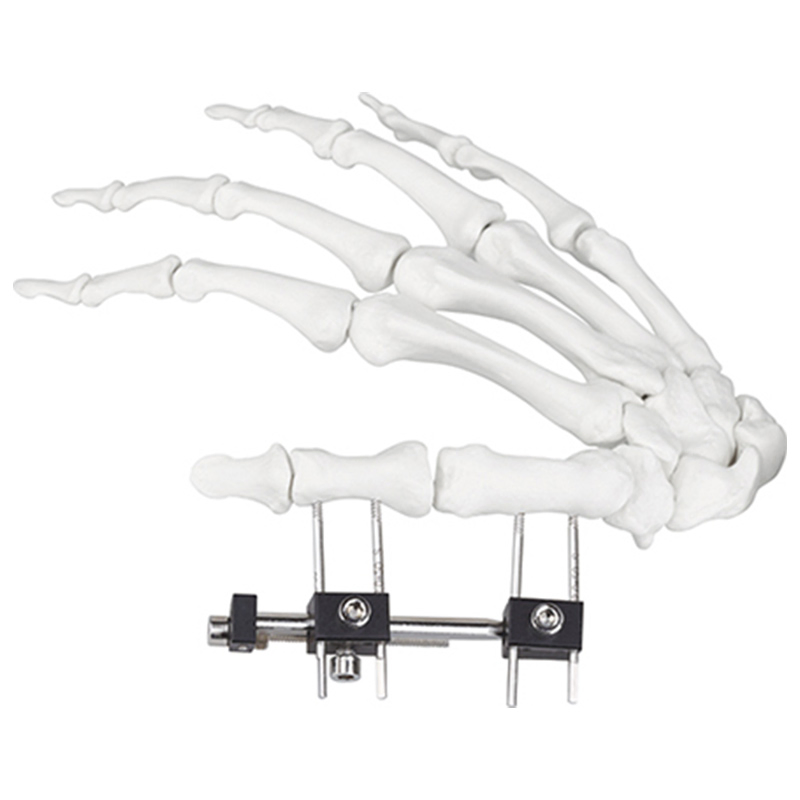
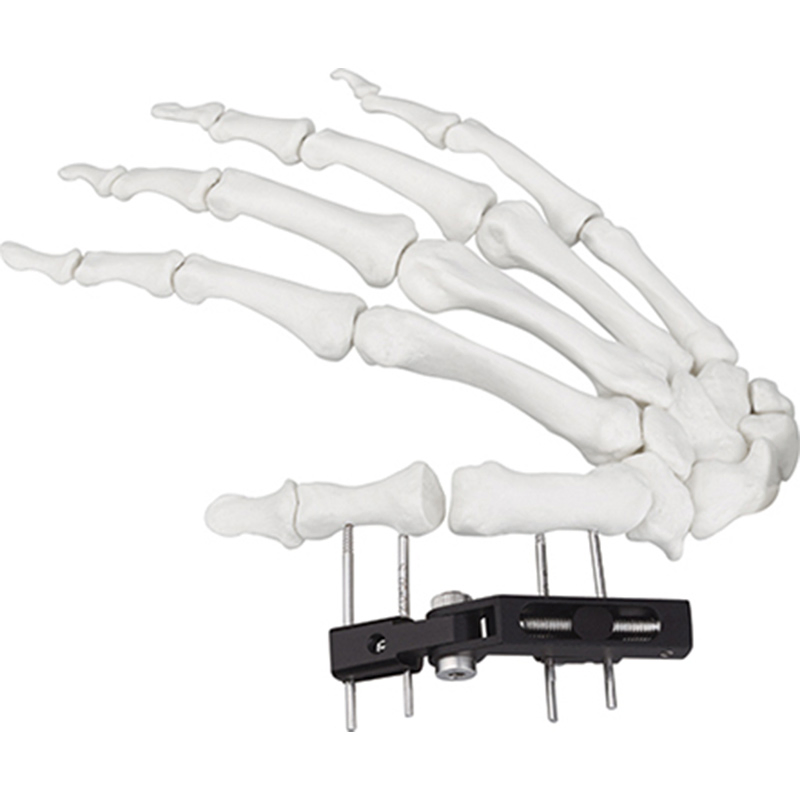
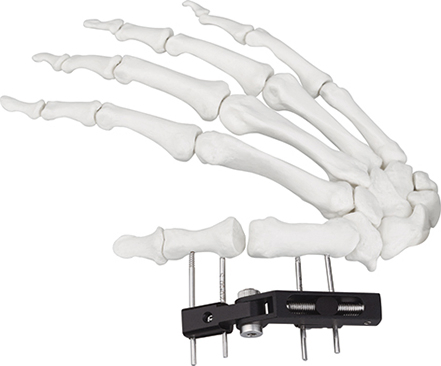
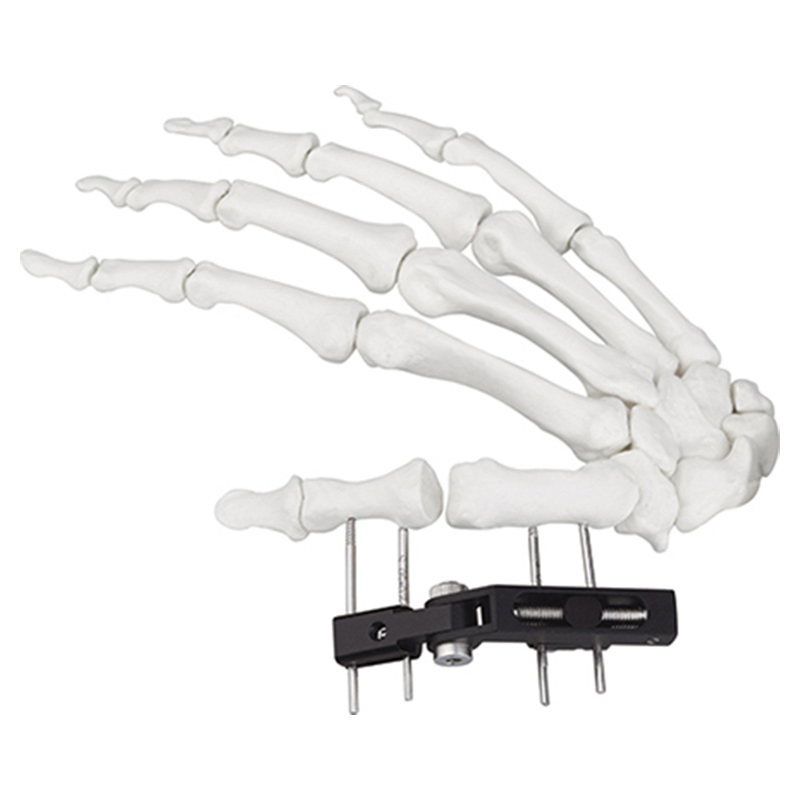
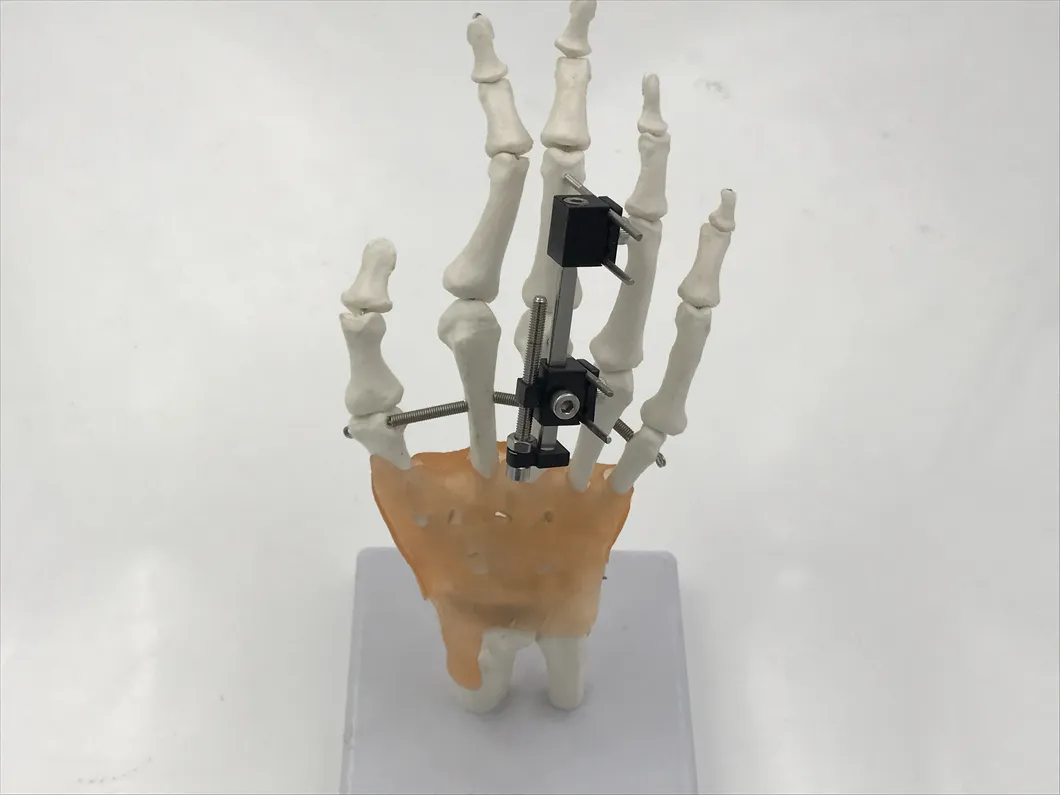
| Product Name | Mini fixator universal compression extension type |
| Specification | Standard screw: φ2.5*50mm 4pcs; 3mm hex wrench, 2mm screwdriver |
| Material | AL (Aluminum) |






Established in 1958, our enterprise is dedicated to the development, manufacture, and sales of orthopedic implants and instruments. As a key enterprise for national medical apparatus and a director company of the industry association, we offer a comprehensive product line including spine pedical screws, interlocking nails, locking plates/screws, trauma plates, cannulated screws, external fixators, and hip prosthesis.
All medical apparatuses have passed the approval and authentication of national and provincial superintendent offices. Our facility features advanced equipment including imported CNC machines and high-precision digital control machines, along with a 100,000 Grade axenic purification chamber to ensure high-quality standards. We were the first to pass ISO 9001 quality system certification and our products have received numerous national invention prizes and science and technology awards.


