




1 / 5










| Model NO. | TY-CS9 |
| Color | Silver |
| Application | Foot and Ankle |
| Material | Pure Titanium |
| Properties | Implant Materials & Artificial Organs |
| Instrument Classification | Class III |
| Warranty | One Year |
| After-Sale Service | Online Technical Support |
| Package | Sterile/Non-Sterile Package |
| Certificate | CE/ISO 13485 |
| OEM | Accepted |
| HS Code | 9021100000 |
| Production Capacity | 10000pieces/Year |
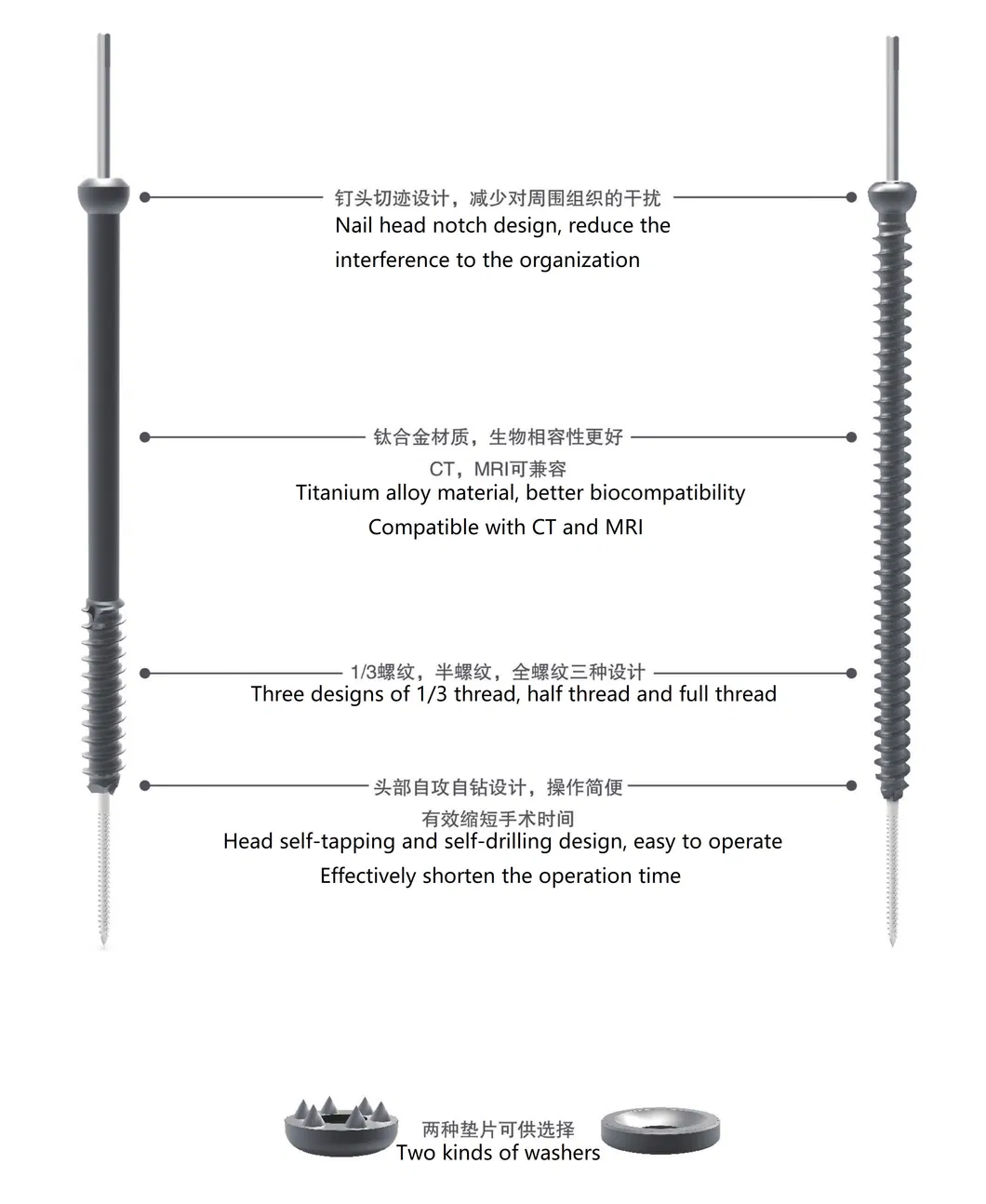
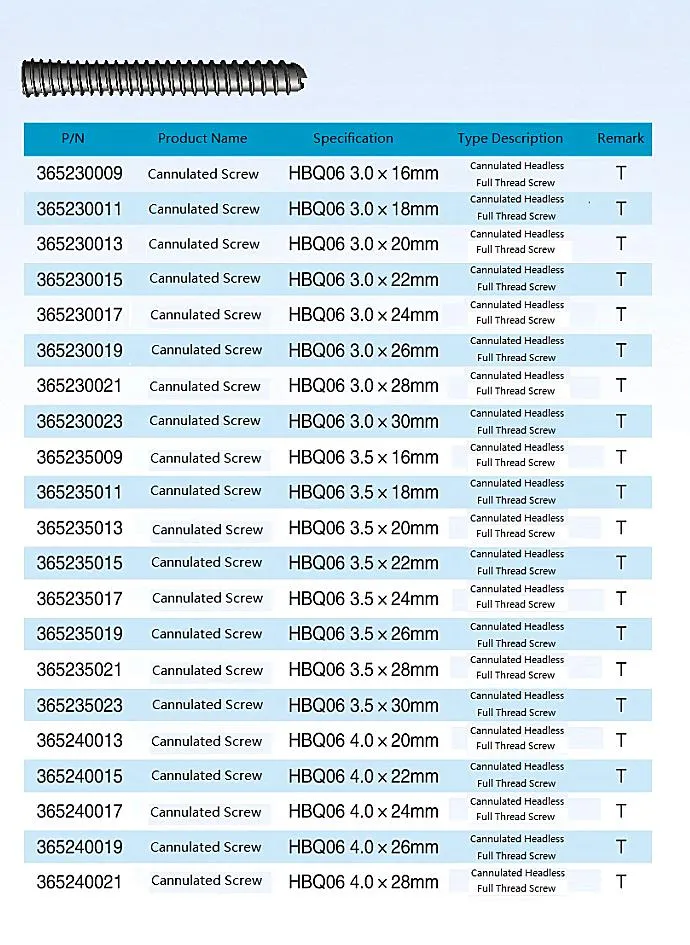
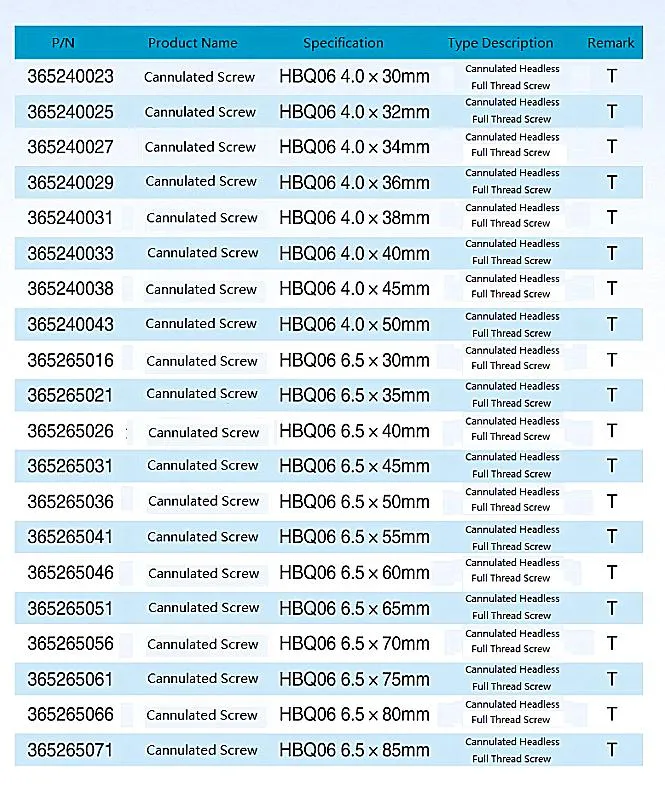



| Item | Value |
| Properties | Implant Materials & Artificial Organs |
| Model Number | hbq |
| Licence Number | CE; ISO13485 |
| Instrument Classification | Class III |
| Material | Titanium |
| Product Name | Cannulated Bone Screw |
| Usage | Orthopedic Surgery |
| MOQ | 1PC |
| Size Range | 14-120mm |
Our pricing strategy reflects the balance between high-grade product quality and market competitiveness. We aim for a Win-win partnership, ensuring that quality is never compromised for price.
*All prices are for reference. Exact price will be based on the latest quotation.



