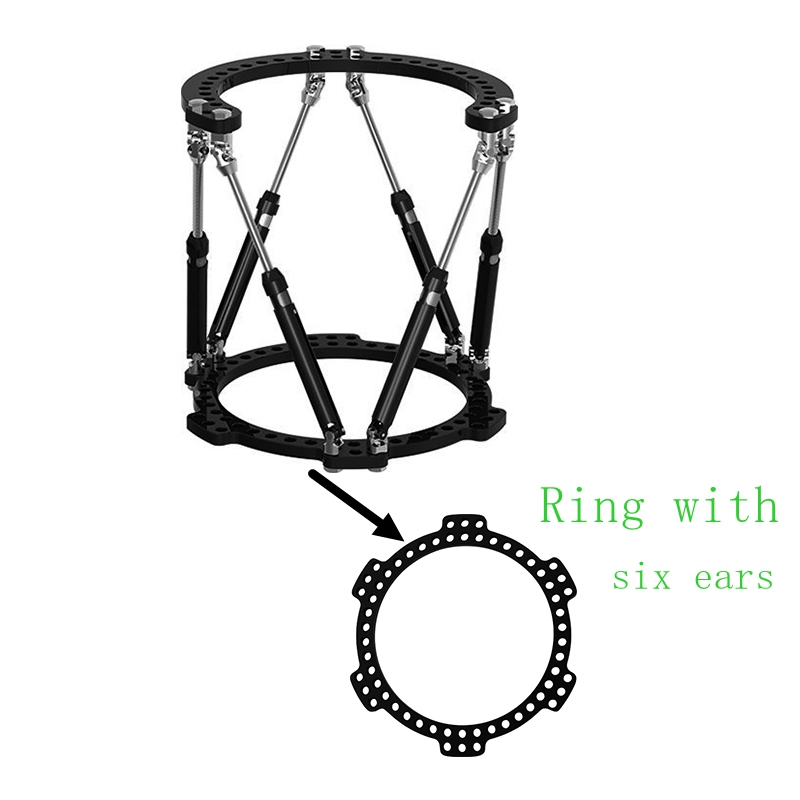
Our Advantages
◈ Annular design, firm and reliable
◈ Easier operation & Shorter surgical time
◈ Minimally invasive surgery, preserves bone blood supply
◈ No second surgery needed for removal
◈ Dynamic design optimized for bone healing
◈ Taper bone screws for taut and firm fixation
◈ Versatile for all kinds of deformity correction
Manufacturer Profile
Established in 1958, the manufacturer is specialized in the development, manufacture, and sales of orthopedic implants and instruments. As a key enterprise for national medical apparatus, the product line covers spine pedicle screws, interlocking nails, locking plates/screws, trauma plates, cannulated screws, external fixators, hip prosthesis, and surgical instruments.
The products are characterized by multi-varieties and complete specifications. Equipped with advanced technology including imported CNC, high-precision digital control machines, and a 100,000 Grade axenic purification chamber, high quality is ensured. The company was the first to pass ISO 9001 quality system certification, with products winning multiple national and provincial awards for innovation and technology.
Frequently Asked Questions
What are the primary products available?
The main product range includes Metal Bone Plates and Screw Systems, Metallic Cannulated Bore Screws, Metallic Interlocking Intramedullary Nails, Spinal Fixation Devices, and External Fixation Devices.
How is product quality guaranteed?
All products are produced according to CE standards. Quality is maintained through pre-production samples before mass production and a final rigorous inspection before shipment.
What is the experience level of the manufacturer?
The manufacturer has over 60 years of experience (established in 1958) in R&D, manufacturing, and the global sale of orthopedic implants and instruments.
What certifications do the products hold?
The facility and products are certified with ISO 9001, CE, and ISO13485, ensuring compliance with international medical device standards.
What payment and delivery terms are accepted?
Accepted delivery terms include FOB, CIF, EXW, and Express Delivery. Payment can be made via T/T, L/C, Western Union, or Escrow in USD, EUR, or CNY.
Can you provide OEM/ODM services?
Yes, OEM and ODM services are accepted to meet specific clinical or regional requirements for orthopedic fixation devices.