📋 Basic Information
Transport Package
PE Inner Bag + PE Outer Bag + Carton
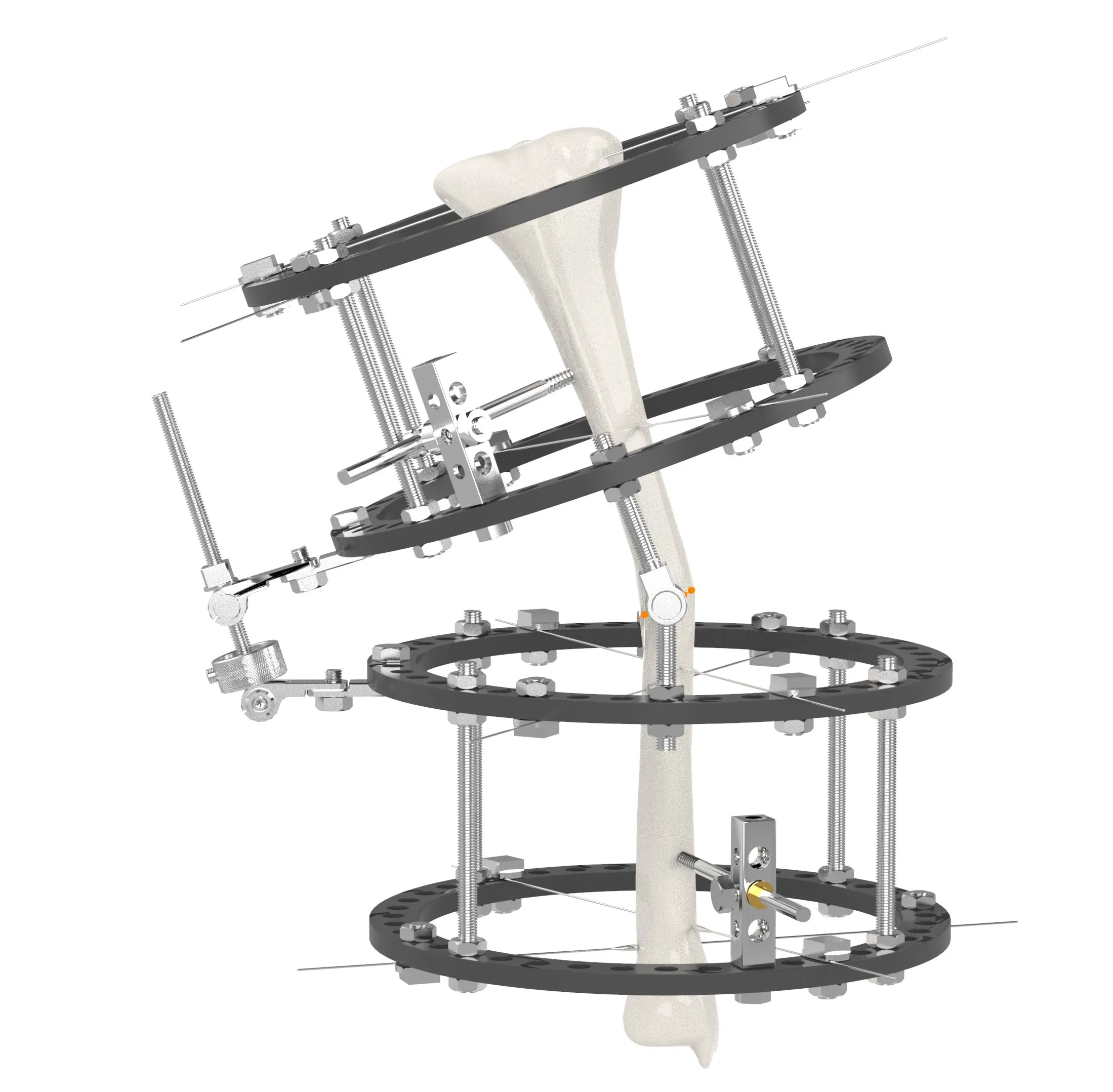
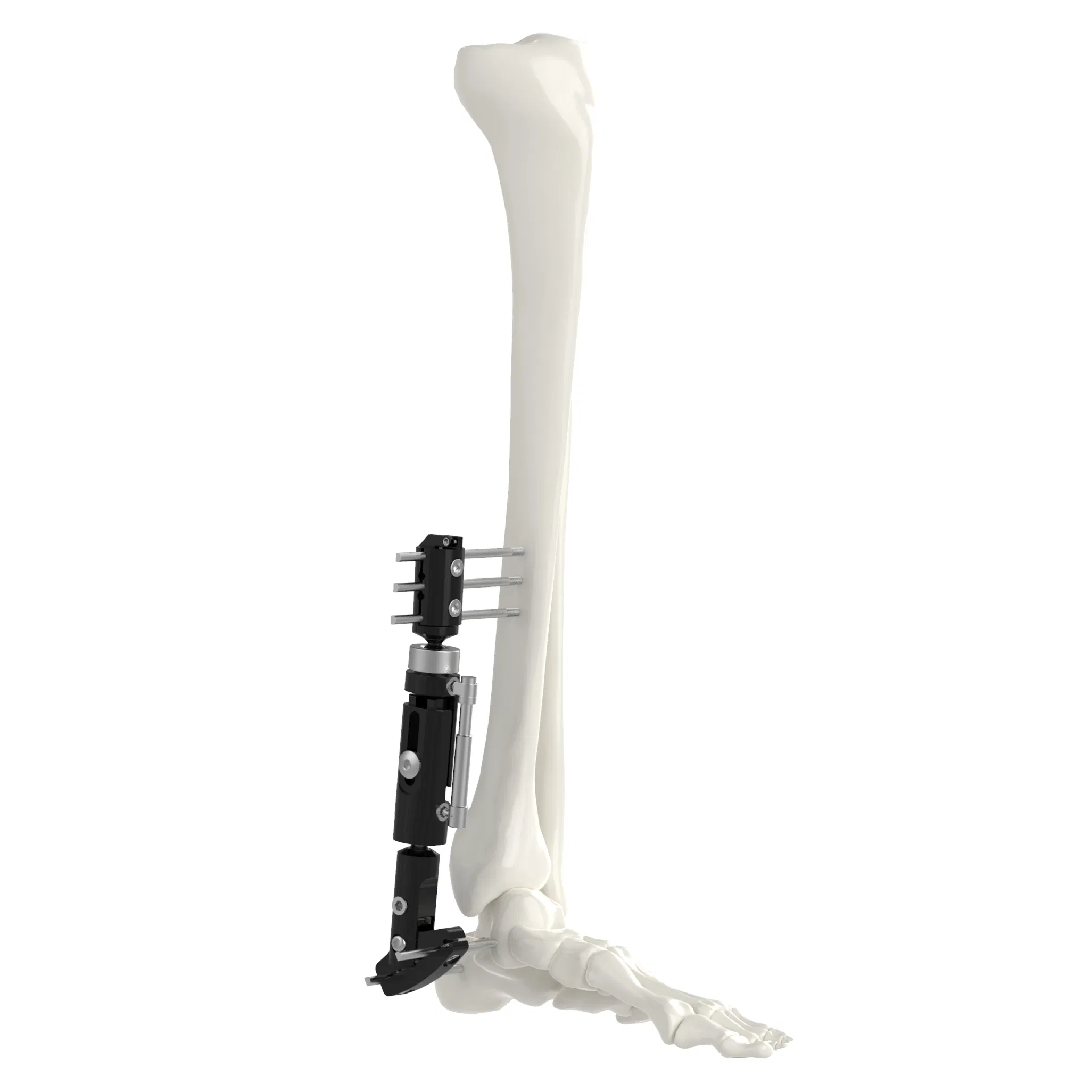
📦 Product Description
| Fixation System Types |
| Single Arm |
Combine |
Ilizarov |
Needle |
🏭 Profile Overview
Manufacturing Excellence
A medical device manufacturer founded in 1994, now recognized as one of the largest designers and suppliers of orthopedic implants and instruments. Operating within a workshop exceeding 60,000 square meters, the facility provides comprehensive production for traumatology, spinal fixation systems, and surgical instruments. Product lines include femoral nails, titanium plates, screws, pedicle screws, PEEK cages, and balloon kyphoplasty systems, serving markets across Europe, Latin America, the Middle East, and Asia.
❓ Frequently Asked Questions
What are the available shipping modes and associated charges?
Orders are typically dispatched via professional courier services or standard post based on client requirements.
Does the facility hold international quality certifications?
Yes, the company is ISO 9001:2008 and EN ISO 13485:2012 certified. A significant portion of the product range also carries the CE mark for compliance with health and safety standards.
What is the minimum order requirement?
Standard minimum orders are expected to be at least US$1000. However, smaller sample orders are accommodated for initial market testing and quality evaluation.
Can customized products be developed if they are not in the current catalogue?
Customization is possible by providing detailed pictures or physical samples. Please note that customized production cycles may require additional lead time.
Are the listed products maintained in regular stock?
Most listed products are produced monthly and kept in stock. Inventory levels fluctuate based on sales conditions, so it is recommended to verify specific availability during the inquiry process.
What specific medical applications are these implants designed for?
These implants are primarily designed for orthopedic trauma surgery, including bone fixation, spinal stabilization, and complex fracture management.