




1 / 5











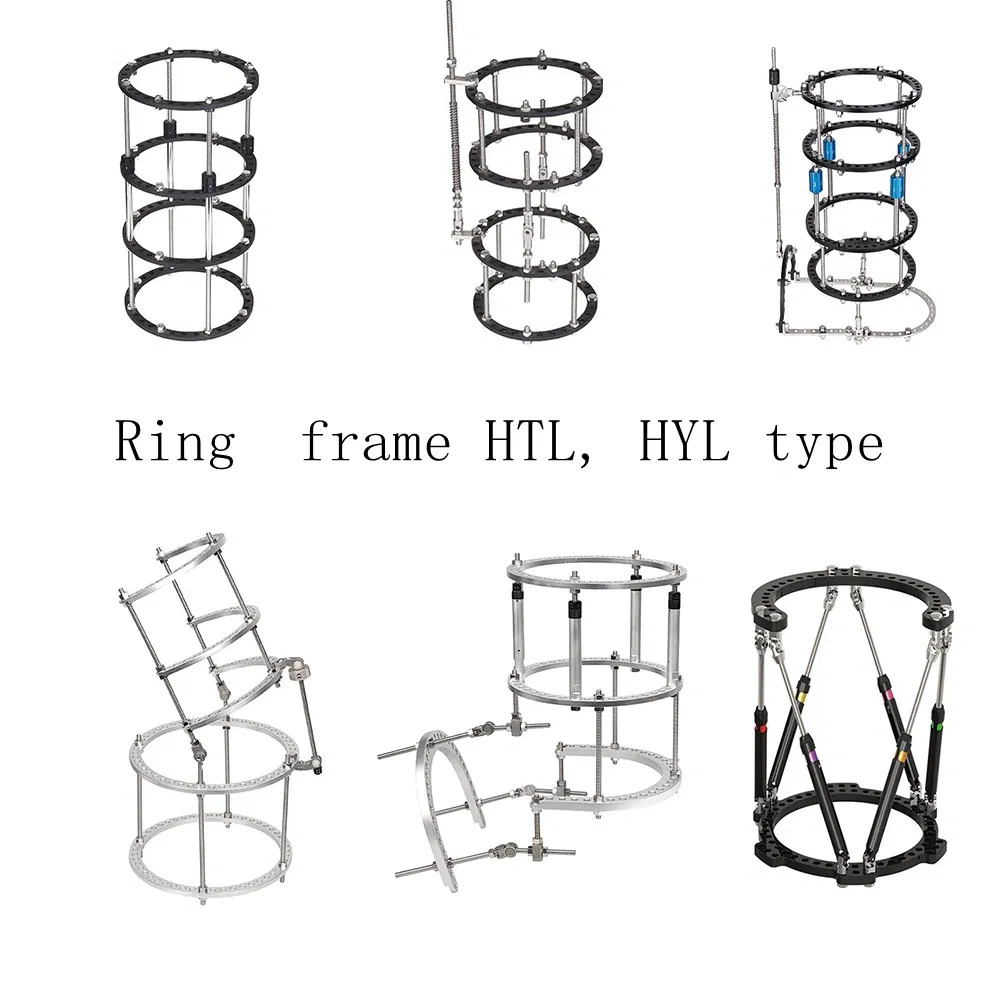
| Name | Number | Size | Material |
|---|---|---|---|
| Foot ring | HTLL04 | L | Aluminum |


Our company was established in 1958 and is engaged in the development, manufacture, and sales of orthopedic implants and instruments. We are recognized as a key enterprise in the national medical apparatus sector. Our product lines cover spine pedicle screws, interlocking nails, locking plates/screws, trauma plates, cannulated screws, external fixators, hip prosthesis, and surgical instruments.
Advanced equipment, including imported CNC machines and high-precision digital control tools, alongside a 100,000 Grade axenic purification chamber, ensures high-quality production. We were among the first to pass ISO 9001 quality system certification, with products winning numerous national and provincial science and technology awards.


