Belgium has long been recognized as a cornerstone of the European MedTech industrial landscape. From the biotechnology clusters in Wallonia to the high-tech engineering hubs in Flanders, the demand for sophisticated Revision Hip/Knee Systems has seen exponential growth. As a leading manufacturer and provider, our reach into the Belgian market is driven by a commitment to clinical excellence and a deep understanding of the local healthcare ecosystem, including renowned institutions like UZ Leuven and CHU de Liège.
In Belgium, the medical device industry is not just a sector; it's an innovation engine. With over 500 companies specialized in medical technology, Belgium serves as a strategic gateway for orthopedic distribution across Europe. Our factories align with the strict EU MDR (Medical Device Regulation) standards required by the Belgian Federal Agency for Medicines and Health Products (FAMHP). This ensures that every hip joint prosthesis and knee replacement system we export meets the uncompromising quality expected by Belgian surgeons.
Belgium’s strategic location, supported by world-class logistics through the Port of Antwerp-Bruges and Liège Airport, allows for rapid delivery of revision arthroplasty components. This logistical efficiency is vital for revision surgeries, which are often more complex and time-sensitive than primary replacements.



A major strength of our company lies in its expertise in 3D printing and customization. In Belgium, a country home to pioneers in additive manufacturing like Materialise, we have adopted similar high-end 3D laser melting technologies to create personalized implants. These systems are designed to address the specific bone loss challenges often found in Belgian patients requiring revision surgery. By creating patient-specific acetabular cups and femoral stems, we enhance treatment outcomes and significantly improve long-term patient comfort.
Utilizing AI algorithms to simulate load distribution in revision knee systems, ensuring longevity in active patients.
Our S-ROM Titanium Alloy systems provide cementless solutions for complex revision arthroplasty, a preferred choice in Belgium.
Fully certified with ISO 13485 and CE Marking, ensuring seamless integration into the Belgian healthcare system.
The Belgian orthopedic market is currently seeing a shift toward minimally invasive revision procedures. As the population ages, the frequency of revision surgeries for primary hip and knee replacements performed 15-20 years ago is increasing. Belgian orthopedic surgeons are increasingly seeking modular systems that allow for intra-operative flexibility.
Moreover, there is a growing trend of "Green Arthroplasty" in Belgian hospitals, where factories are pushed to reduce waste. Our manufacturing process incorporates sustainable material sourcing and optimized sterilization packaging to meet these local ecological expectations.
In Belgian clinical settings, our Revision Hip/Knee Systems are utilized in diverse scenarios:
To date, our products have been widely used in dozens of countries across Asia, Latin America, Africa, and Europe, and have gained recognition from local distributors and surgeons. We offer orthopedic implants, joint implants, brushless motors, and other high-precision medical products tailored for the Belgian market.
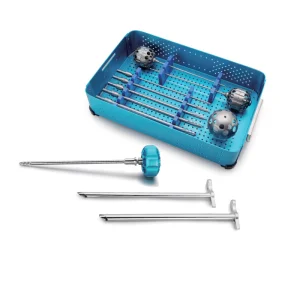 Orthopedic Endoscope Instruments Set Surgical Instrument Bone Surgery
Orthopedic Endoscope Instruments Set Surgical Instrument Bone Surgery
 S-ROM Titanium Alloy Cementless Hip Joints for Revision Arthroplasty Surgery
S-ROM Titanium Alloy Cementless Hip Joints for Revision Arthroplasty Surgery
 Orthopedic Toolmed Knee Prosthesis Manufacturer Knee Replacement Ortho Surgery Arthroplasty Tka
Orthopedic Toolmed Knee Prosthesis Manufacturer Knee Replacement Ortho Surgery Arthroplasty Tka
 Total Hip and Knee Joint Replacement Metal Titanium Implant Made in China
Total Hip and Knee Joint Replacement Metal Titanium Implant Made in China
 Disposable Pulsed Lavage System
Disposable Pulsed Lavage System
 Orthopedic Surgical Mis Minimally Invasive Spine Pedicle Screw Instrument Set
Orthopedic Surgical Mis Minimally Invasive Spine Pedicle Screw Instrument Set
 Surgical Instrument Femoral Tibial Intertan Nail Instrument Set (Type 1) Medial Equipment
Surgical Instrument Femoral Tibial Intertan Nail Instrument Set (Type 1) Medial Equipment
 Replacement CE Sterilized, Carton Package Prosthesis Knee Joint
Replacement CE Sterilized, Carton Package Prosthesis Knee Joint
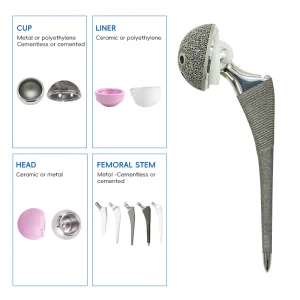 UHMWPE Insert Acetabular Cup Hip Joint Hemiarthroplasty System Total Knee Replacement Prosthesis Implants Instruments Set
UHMWPE Insert Acetabular Cup Hip Joint Hemiarthroplasty System Total Knee Replacement Prosthesis Implants Instruments Set
 Orthopedic Replacement Sterilized Package Prosthesis Knee Joint
Orthopedic Replacement Sterilized Package Prosthesis Knee Joint
 Factory Price Quality Assured Pfna Nail Instrument Set
Factory Price Quality Assured Pfna Nail Instrument Set
 Locking Compression Plate (LCP) for Femur - LC-LCP Orthopedic Implant System for Femoral Shaft or Condylar Fracture Surgery
Locking Compression Plate (LCP) for Femur - LC-LCP Orthopedic Implant System for Femoral Shaft or Condylar Fracture Surgery
Join the network of elite Belgian hospitals and distributors who trust our precision-engineered revision systems.