🦾
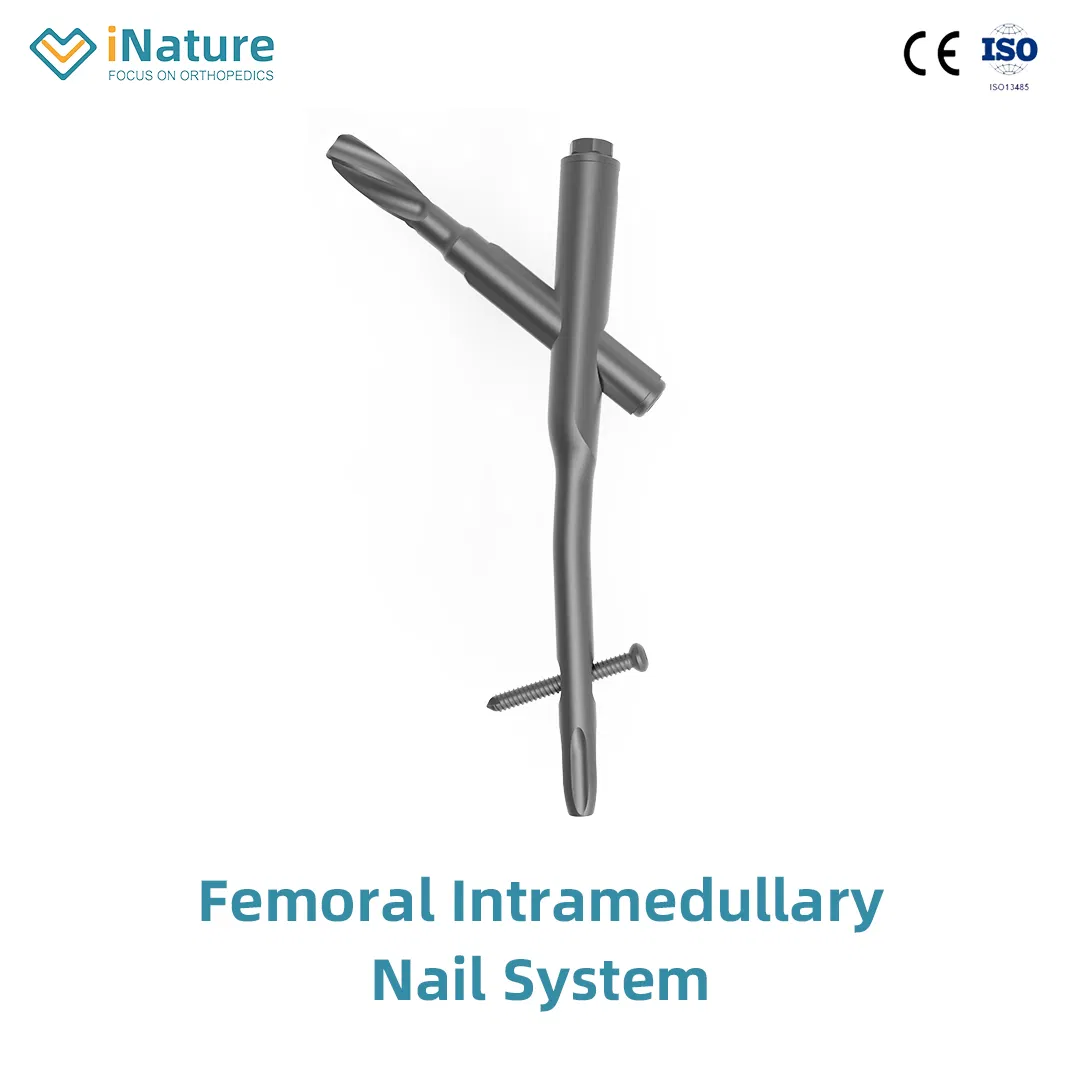
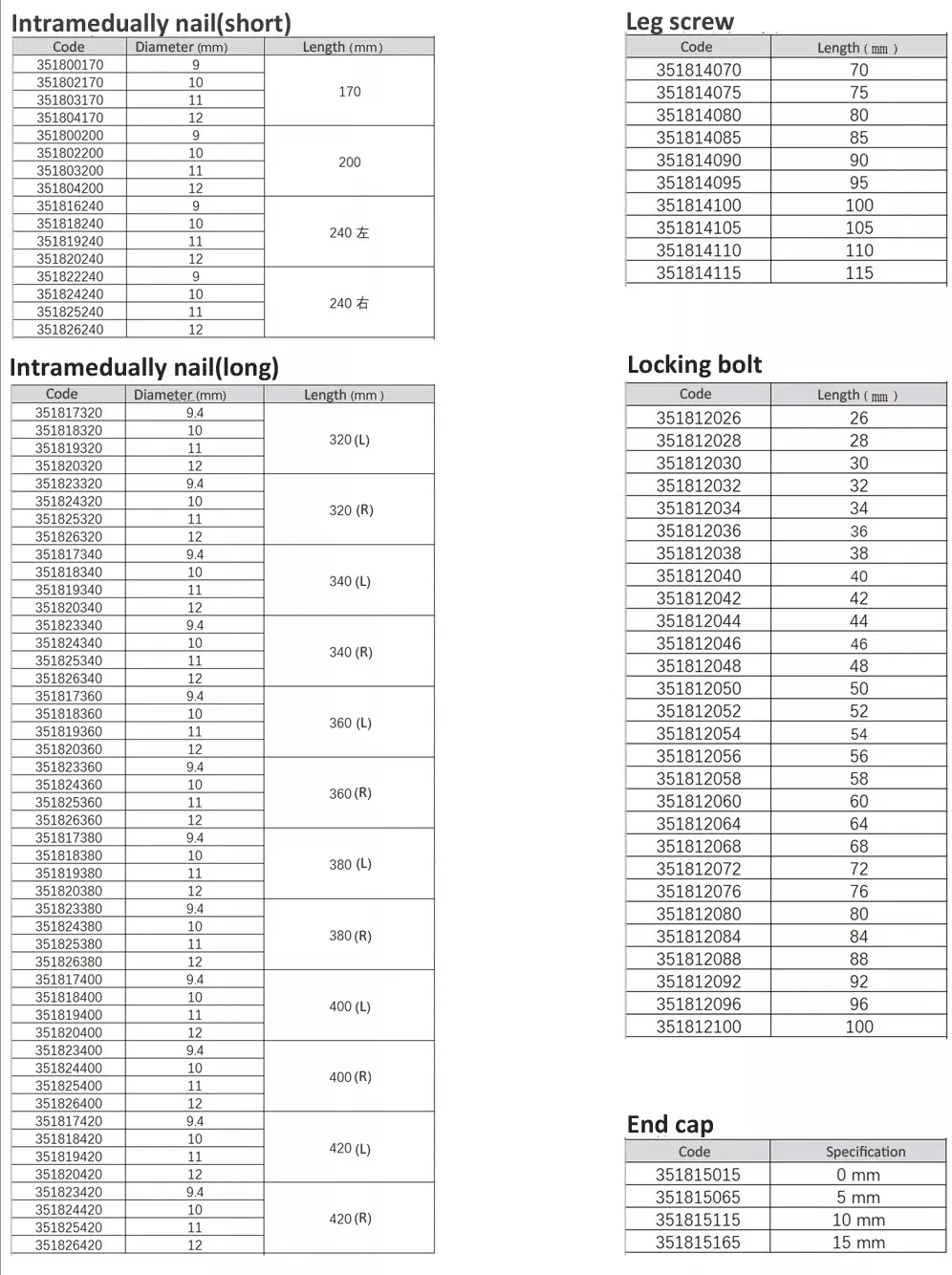
Femur Intramedullary Nail System
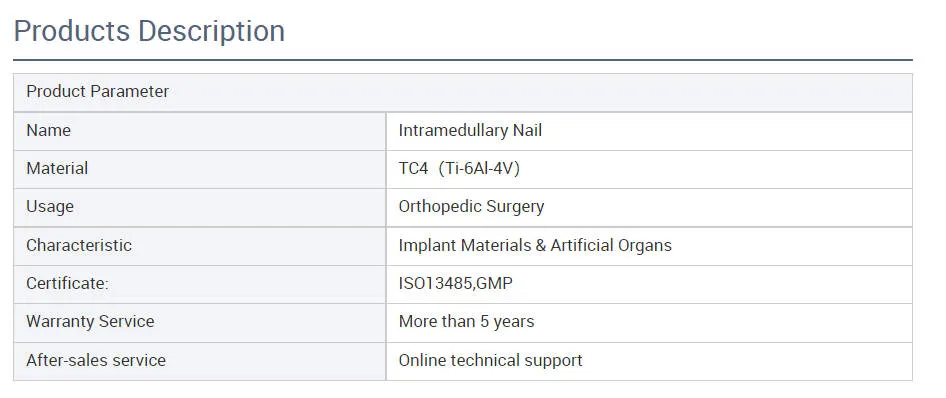
Material: Titanium / Surface Finished: Oxidation & Milling
Anatomical Design: Medial-lateral angle of 5° allows insertion at the tip of the greater trochanter.
Stress Reduction: Lateral flatten design eases insertion and reduces stress to the lateral cortical bone.
Elastic Groove Tip: Eases insertion and reduces concentration stress.
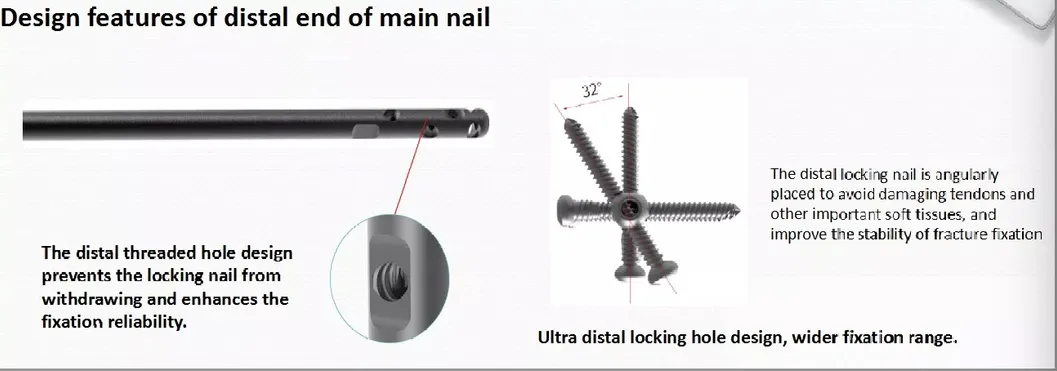
Curved Design: 240mm distal curved design prevents nail tip contact with cortical bone, reducing resistance and pain.
Lag Screw Stability: Provides excellent compaction to cancellous bone, ideal for osteoporotic patients.
High Efficiency: Locking bolt with double thread design to reduce operation time.
❓
Frequently Asked Questions
1. What is the material used for these intramedullary nails?
The system is primarily made of Pure Titanium, which offers excellent biocompatibility and strength for trauma and fracture fixation.
2. What kind of warranty and guarantees do you provide?
We offer a one-year warranty. This includes a guarantee for return and exchange within one month for quality issues (non-operational errors) and free repairs within the first year.
3. Is the product certified for global medical use?
Yes, the products carry CE and ISO 13485 licenses, meeting Class III instrument classification standards.
4. Can I customize the products through OEM?
Yes, OEM services are accepted to meet specific surgical or institutional requirements.
5. How does the design reduce patient pain?
Features like the 240mm distal curved design and elastic groove tip help prevent the nail from pressing against the cortical bone, reducing insertion resistance and pain incidence.
6. How are the products packaged for transport?
All products are provided in a Sterile Package to ensure safety and readiness for clinical use upon delivery.