Frequently Asked Questions
Q1: What materials are used in the construction of the external fixators?
The system is constructed using high-grade Titanium Alloy and Stainless Steel for durability, alongside medical-grade carbon fiber for optimal radiolucency.
Q2: Are these external fixation systems reusable?
Yes, the system is designed with reusability in mind, making it a sustainable choice for orthopedic bone fixation procedures.
Q3: Which certifications does the product hold for medical use?
The external fixator system is fully certified with CE and ISO13485 standards, ensuring compliance with international medical safety regulations.
Q4: Can the bone screws be adjusted independently?
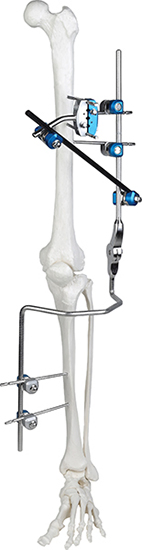
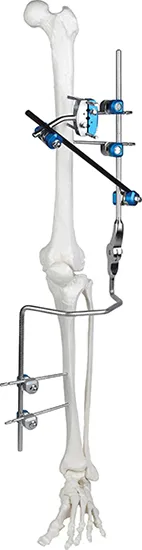
Absolutely. The modular design allows for the independent arrangement of bone screws for both proximal and distal fractures, providing surgeons with maximum flexibility.
Q5: What anatomical areas can this system be used for?
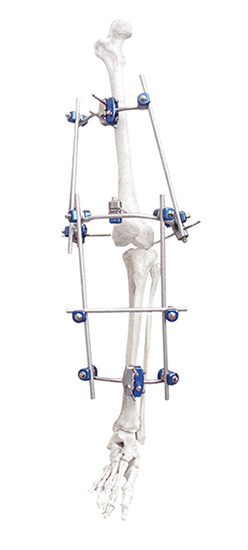
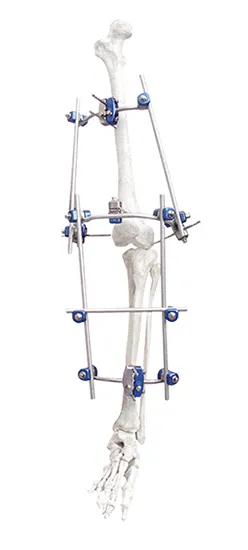
This versatile system includes specialized modules for the Knee Joint, Ankle Joint, Pelvis, and Tibia/Perone fractures.
Q6: What is the benefit of the carbon fiber rods in this system?
Carbon fiber rods provide excellent structural stability while being lightweight and X-ray transparent, which significantly enhances fracture visualization during follow-up.