
1 / 5












| Product | Size / Length (mm) | Material |
|---|---|---|
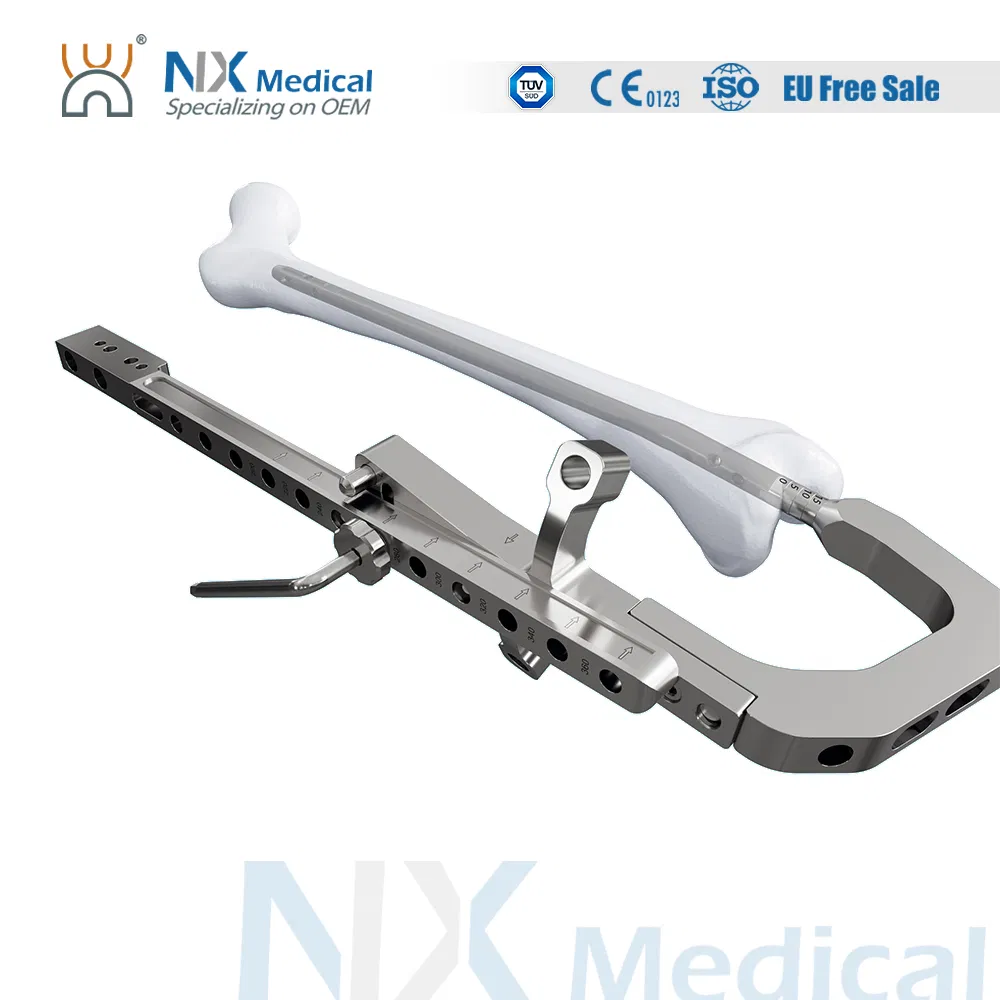
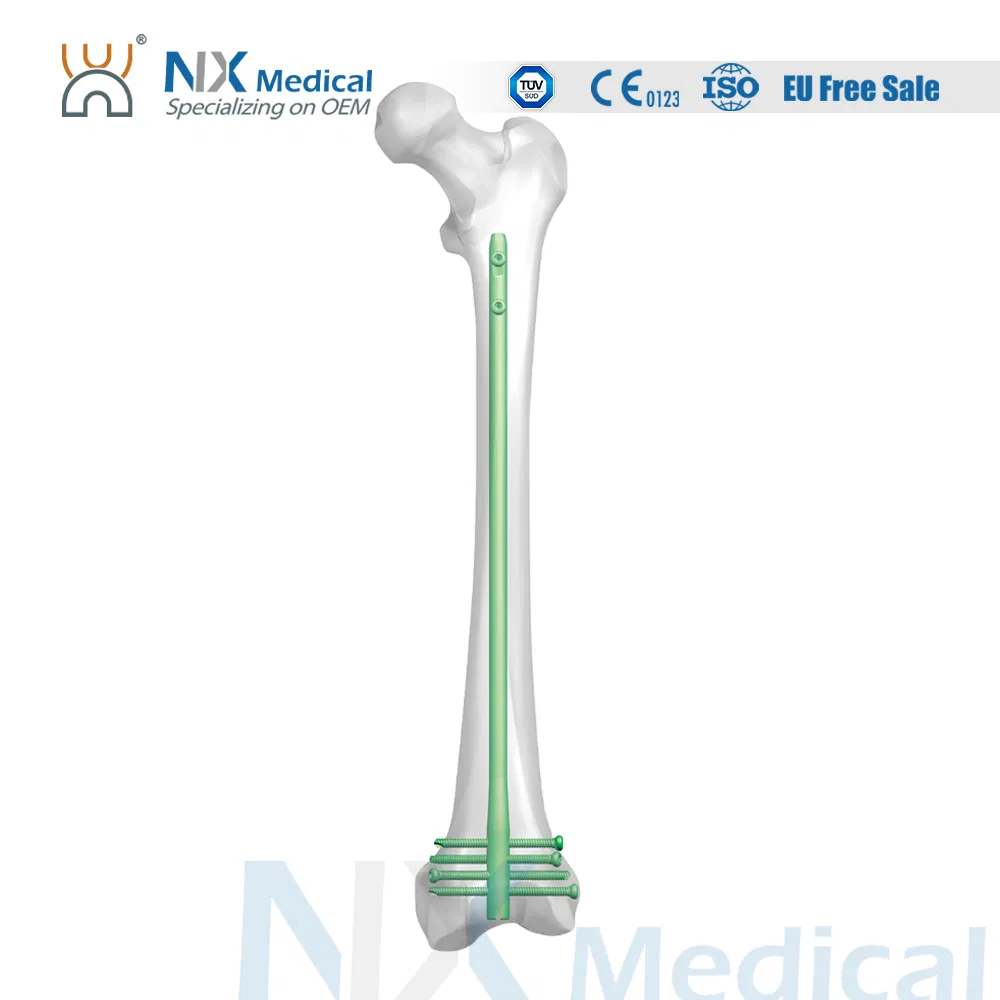
| Retrograde Femoral Nail | 9 x 200 / 220 / 240 / 260 / 280 / 300 / 320 / 340 / 360 | Titanium Alloy |
| 10 x 200 / 220 / 240 / 260 / 280 / 300 / 320 / 340 / 360 | Titanium Alloy | |
| 11 x 200 / 220 / 240 / 260 / 280 / 300 / 320 / 340 / 360 | Titanium Alloy | |
| 12 x 200 / 220 / 240 / 260 / 280 / 300 / 320 / 340 / 360 | Titanium Alloy | |
| Interlocking Screw (φ4.8mm) | 26 / 28 / 30 / 32 / 34 / 36 / 38 / 40 / 42 / 44 / 46 / 48 / 50 / 52 / 54 / 56 / 58 / 60 / 64 / 68 / 72 / 76 / 80 / 84 | Titanium Alloy |
| End Cap (Extension) | Standard / 5mm / 10mm / 15mm | Titanium Alloy |
We are a leading Orthopedic product manufacturer located in East China, providing a comprehensive range of cost-effective surgical and reliable therapeutic solutions to orthopedic surgeons and patients worldwide. Our portfolio includes superior trauma implants, spinal implants, and surgical instruments. Committed to global innovation, we invest heavily in R&D to maintain a competitive position in the international market.
Our major categories include locking plates, intramedullary nails, cannulated headless screws, and spinal systems such as cervical plates and pedicle screws. All products are certified by TUV CE0123 and NMPA, complying with ISO13485 quality standards to ensure the highest safety for medical procedures.





