Basic Information
Sterilization
Without Ethylene Oxide Sterilization
Quality Guarantee
Two Years
Customization
Non-Customized
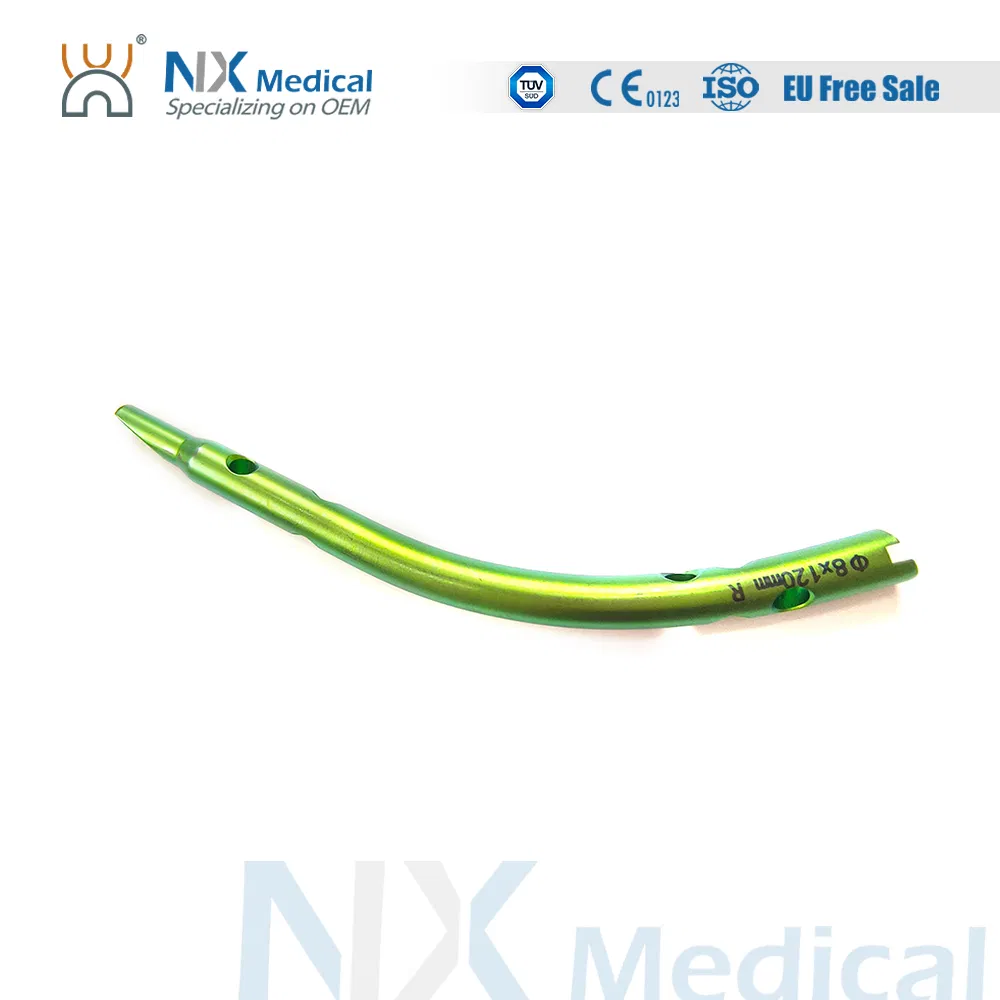
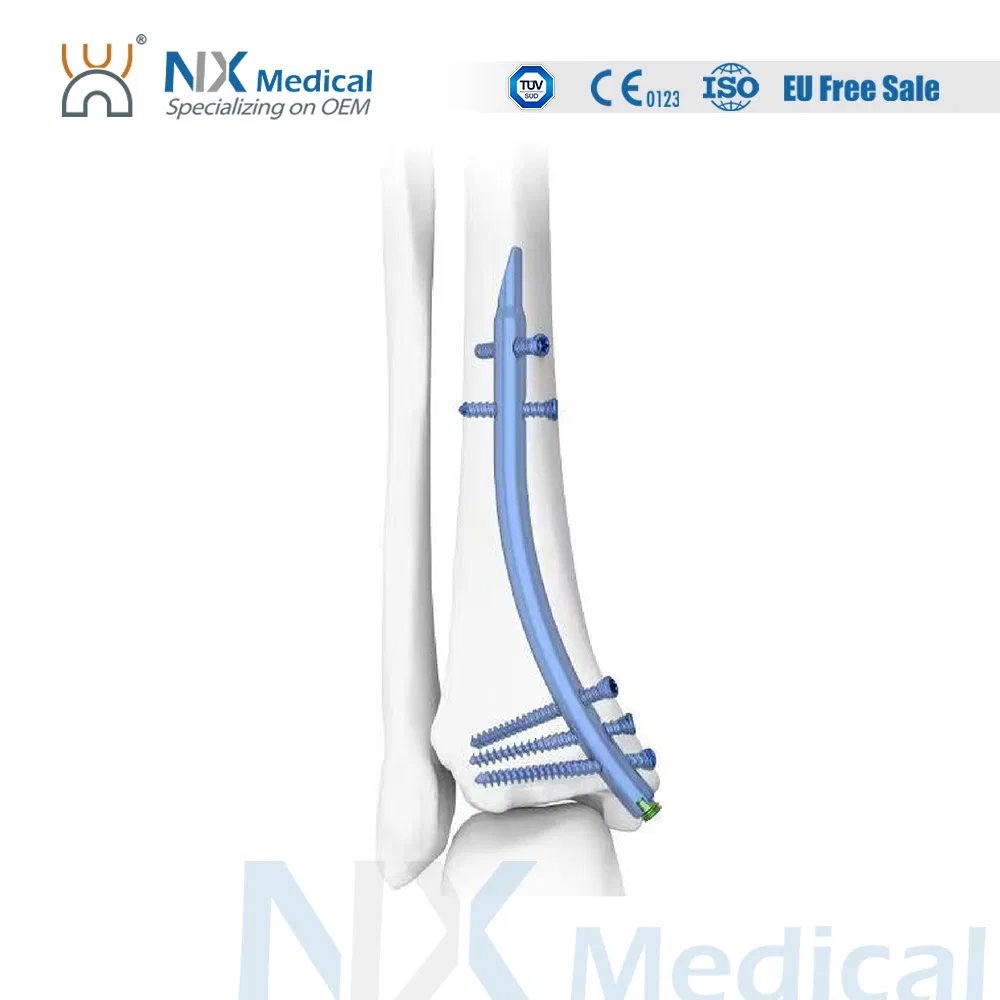
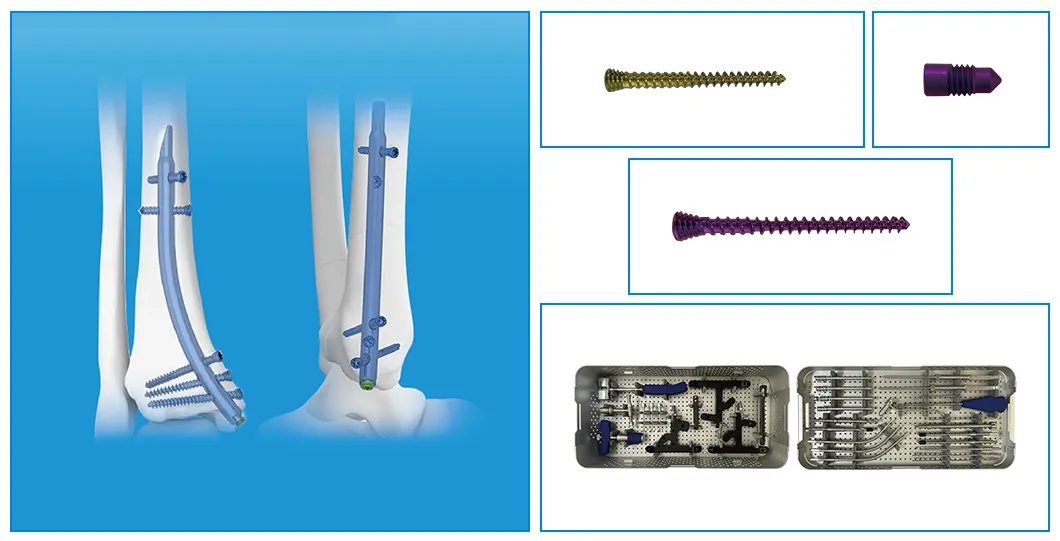
Retrograde Tibial Nail System
Key Features and Benefits:
01
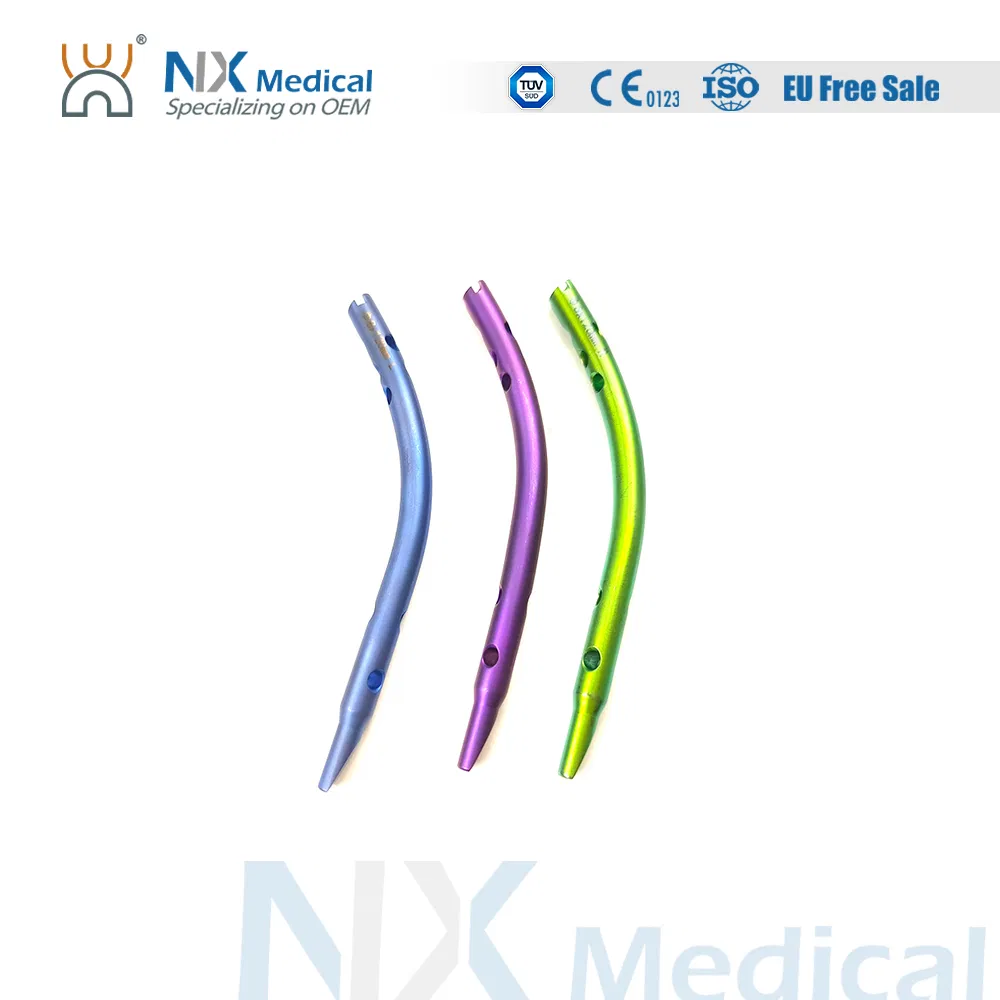
Colour coding to easily differentiate nail sizes.
02
Proximal oblique cutting design of the nail reduces irritation to soft tissues of medial malleolus.
03
Two proximal interlocking screws avoid fracture rotation and displacement.
04
Distal flat cutting design of the nail avoids the insertion difficulties.
05
Three distal divergent locking screws provides effective support and fixation.
06
Advanced and user-friendly instruments facilitate nail insertion.
07
Carbon fiber jigs facilitate first distal screw insertion position preparation.
Product Specifications
| Product |
Size / Length (mm) |
Material |
| Retrograde Tibial Nail |
7 x 110 |
TA (Titanium Alloy) |
| 8 x 120 |
TA |
| 8 x 140 |
TA |
| φ3.5mm Interlocking Cancellous Screw |
30 / 32 / 34 / 36 / 38 / 40 / 42 / 44 / 46 / 48 / 50 / 52 |
TA |
| φ3.5mm Interlocking Cortical Screw |
18 / 20 / 22 / 24 / 26 / 28 |
TA |
| φ4.0mm Interlocking Cortical Screw |
24 / 26 / 28 / 30 / 32 / 34 |
TA |
| Retrograde Tibial Nail End Cap |
Standard / 5 |
TA |
Manufacturing & Quality
Our facility is a leading Orthopedic product manufacturer located in East China, providing comprehensive, cost-effective surgical and reliable therapeutic solutions to surgeons and patients worldwide. We offer a wide range of trauma implants, spinal implants, and high-precision surgical instruments.
We believe superior quality is the key for patient recovery. Internally, we have established a strict quality system for frequent self-audit. Externally, we comply with ISO13485, and our products are certified by TUV CE0123 and NMPA.
Our goal is to provide the safest and most advanced Orthopedic implants and technologies to ensure reliable operating procedures, rapid recovery, and a pain-free life after surgery. We also act as a major OEM supplier to large global players in the industry, maintaining long-term cooperation strategies with world-famous orthopedic firms.
Frequently Asked Questions
Can I request a sample order?
Yes, we welcome sample orders to test and check quality. Mixed samples are acceptable.
Do you accept customized product requests?
Yes, we have a complete design and drawing department along with CNC machinery to provide high-precision customized services.
How do I proceed with an order?
Firstly, let us know your requirements. We then quote accordingly. Once confirmed, the customer places a deposit, and we arrange production.
What is the typical sample lead time?
For existing items, it takes within 3 days. For OEM products, the process takes approximately 2 weeks.
Can you brand the products with our logo?
Yes, we can print your logo on both products and packaging, provided the Minimum Order Quantity (MOQ) requirements are met.
How is product quality ensured before shipment?
We conduct strict sampling inspections on all products before shipment and ensure intact, secure packaging for transit.