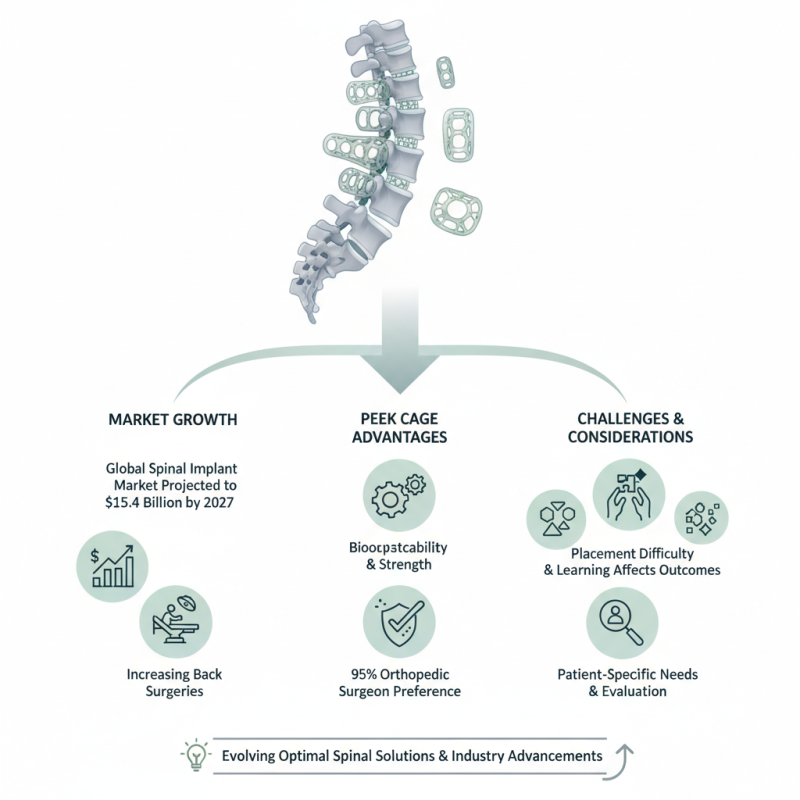
The demand for TLIF PEEK Cage solutions has grown significantly in recent years. The global spinal implant market is projected to reach $15.4 billion by 2027, driven by increasing back surgeries. TLIF PEEK Cages are known for their biocompatibility and strength. Studies show that 95% of orthopedic surgeons prefer these cages for spinal fusion procedures.
Despite their popularity, challenges remain. Surgeons report difficulties with cage placement and the learning curve associated with new techniques. Additionally, the variability in cage designs can affect outcomes, leading to further analysis. It's crucial to consider the specific needs of each patient, as no single cage design is perfect for every case.
As we explore the best TLIF PEEK Cage options for 2026, understanding these nuances is vital. Selecting the right cage involves more than just price; it requires careful evaluation of each product's features and clinical data. The journey toward optimal spinal solutions continues to evolve, reflecting both advancements and areas for improvement in the industry.
Transforaminal Lumbar Interbody Fusion (TLIF) is a surgical procedure aimed at treating various spinal conditions. It involves removing damaged disc material and stabilizing the spine with an interbody cage. The TLIF technique minimizes muscle disruption, which can lead to quicker recovery times for patients. Surgeons often prefer this technique for its effectiveness in alleviating back pain.
The role of PEEK (Polyether Ether Ketone) cages is essential in TLIF procedures. These cages are made from a biocompatible material known for its durability and strength. PEEK cages promote fusion by providing a stable environment for bone growth. Their radiolucent nature allows for clear imaging during follow-up assessments. Many surgeons appreciate how PEEK does not interfere with X-rays. However, it is important to note that not all PEEK cages are created equal. Variations in design and surface texture can affect fusion outcomes.
As the demand for TLIF procedures grows, so does the need for reliable cage options. Surgeons must consider factors like cage size and geometry. Some designs may not fit all patients. More research is needed to identify the best choices. Individualized approaches to spinal care can lead to better results. It is crucial for healthcare providers to stay informed about advancements in this area.
PEEK cages have gained popularity in TLIF (Transforaminal Lumbar Interbody Fusion) surgeries. These devices are made from a biocompatible polymer known for its excellent mechanical properties. Surgeons appreciate PEEK cages because they exhibit stiffness similar to bone. This provides a stable environment for spinal fusion. Additionally, PEEK is radiolucent, meaning it does not interfere with imaging techniques like X-rays. This feature aids in post-operative evaluations.
Another significant benefit of PEEK cages is their resistance to wear and corrosion. Unlike metal alternatives, PEEK does not create artifacts in imaging. This makes it easier to monitor healing progress. PEEK also integrates well with biological tissues, promoting osseointegration. However, some studies suggest that the lack of material stiffness can be a double-edged sword. While it mimics bone well, some doctors report concerns about its long-term stability in certain patients.
Surgeons need to weigh the pros and cons carefully. Choosing the right interbody cage requires not only understanding the material but also patient-specific factors. Each procedure is unique. Therefore, ongoing research and clinical feedback are crucial for optimal outcomes. As medical technology evolves, evaluating all options will help ensure the best possible care for patients.
In 2026, the market for PEEK TLIF cages is expected to grow significantly. According to a recent study by Grand View Research, the global spinal implant market, including TLIF cages, is projected to reach USD 17.6 billion by 2027. This growth highlights the increasing demand for advanced alternatives in spinal fusion surgery.
Top manufacturers are focusing on innovative designs and materials. PEEK, or polyether ether ketone, is favored for its excellent biocompatibility and mechanical strength. Reports show that PEEK's modulus of elasticity closely matches that of human bone, making it a suitable choice for spinal implants. This alignment with natural tissue reduces the risk of subsidence, a common issue seen in traditional metallic cages.
However, challenges remain. Many providers still face difficulties in educating surgical teams about the benefits and proper use of PEEK cages. There is a need for more clinical studies to validate performance differences. As the industry evolves, experienced manufacturers must address these knowledge gaps, ensuring that surgeons can make informed decisions for patient care. The shift to PEEK cages represents hope, but awareness and training are crucial for maximizing their benefits.
The evolution of PEEK (polyetheretherketone) cage designs has significantly impacted spinal surgery. Recent studies show PEEK cages provide favorable biomechanical properties compared to traditional materials. For instance, a report by the American Journal of Orthopedics indicates that PEEK cages exhibit a similar elasticity to human bone, reducing stress shielding. This characteristic is crucial for maintaining bone integrity post-surgery.
Innovative designs are pushing the boundaries of PEEK application. Recent data reveal that porous PEEK structures enhance bone ingrowth and stability. A comprehensive meta-analysis noted a 30% increase in fusion rates with these designs. However, challenges remain, particularly in achieving optimal surface roughness for better fixation. Many designs still require improvements in this area to enhance clinical outcomes.
While PEEK's potential is clear, it is essential to acknowledge the gaps in current research. The long-term effects of different PEEK formulations on patient outcomes have not been extensively studied. Thus, careful consideration of cage design and material choice is critical in advancing spinal fusion techniques. Ongoing research is necessary to address these nuances and improve patient care.
The future of PEEK cage technology for TLIF (Transforaminal Lumbar Interbody Fusion) applications is promising. Innovations continue to emerge, aimed at improving spinal stability and patient outcomes. Recent studies indicate that PEEK materials exhibit excellent biocompatibility and mechanical properties. These traits enhance the integration of implants with bone tissue, a crucial factor in successful spinal fusions.
Advancements in the design of PEEK cages are noteworthy. Engineers are exploring porous structures, allowing for better osseointegration. These designs promote the growth of bone into the cage, which may improve stability over time. However, not all designs achieve this goal effectively. Some prototypes have faced challenges related to load-bearing capacity. This highlights the importance of thorough testing and refinement in the development process.
Moreover, the significance of customization is becoming apparent. Personalized implants may address individual anatomical variations, enhancing surgical outcomes. However, the complexity of manufacturing these implants raises questions about cost and accessibility. As we strive for advancements, it's essential to strike a balance between innovation and practicality. Only through continued research can we realize the full potential of PEEK cages in TLIF surgery.
| Model Name | Material Type | Size Options (mm) | Weight (g) | Features | Expected Cost (USD) |
|---|---|---|---|---|---|
| Model A | PEEK | 8x8, 10x10 | 12 | Radiolucent, Bio-compatible | 250 |
| Model B | PEEK | 9x9, 11x11 | 10 | Lightweight, Durable | 300 |
| Model C | Composite PEEK | 10x10, 12x12 | 14 | Enhanced Strength, Lower Wear | 350 |
| Model D | PEEK | 7x7, 9x9 | 9 | Flexible Design, Easy Handling | 200 |
: PEEK cages are used in Transforaminal Lumbar Interbody Fusion (TLIF) surgeries. They provide stability for spinal fusion.
PEEK cages are biocompatible, resistant to wear, and radiolucent. They do not interfere with imaging techniques.
PEEK cages do not create imaging artifacts like metal cages. This allows better monitoring of healing progress.
Some doctors worry about the long-term stability of PEEK in certain patients. Further studies are needed.
Porous PEEK enhances bone ingrowth and stability. This design shows a 30% increase in fusion rates.
Optimal surface roughness is necessary for better fixation. Many current designs need improvements in this aspect.
PEEK exhibits stiffness similar to bone. This characteristic helps maintain integrity after spinal surgery.
Yes, ongoing research is crucial. It helps address gaps in current studies about PEEK formulations’ long-term effects.
Surgeons must consider both material properties and patient-specific factors for optimal outcomes in spinal surgery.
While PEEK mimics bone well, some stiffness concerns may arise. This requires careful evaluation by surgeons.
The article titled "2026 Best TLIF PEEK Cage Options for Global Buyers?" provides a comprehensive overview of TLIF (Transforaminal Lumbar Interbody Fusion) procedures, highlighting the critical role that PEEK (Polyether Ether Ketone) cages play in these surgeries. It discusses the key features and benefits of using TLIF PEEK cages, such as their biocompatibility, mechanical strength, and ability to promote spinal fusion, contributing to improved patient outcomes.
Additionally, the article examines the top global manufacturers of PEEK TLIF cages scheduled for 2026, offering a comparative analysis of different cage designs and materials utilized in the market. The future trends in PEEK cage technology are also explored, emphasizing ongoing innovations aimed at enhancing performance and adaptability in TLIF applications. This analysis serves as a valuable resource for buyers interested in the latest advancements and options available in the realm of TLIF PEEK cages.

 Axton Orthopedic Implants
Axton Orthopedic Implants